Advertisements
Advertisements
प्रश्न
An ideal gas undergoes four different processes from the same initial state (figure). Four processes are adiabatic, isothermal, isobaric and isochoric. Out of 1, 2, 3 and 4 which one is adiabatic.

पर्याय
4
3
2
1
MCQ
Advertisements
उत्तर
2
Explanation:
For curve 4 pressure is constant, so this is an isobaric process.
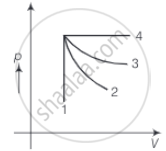
For curve 1, volume is constant, so it is an isochoric process. Between curves 3 and 2, curve 2 is steeper, so it is adiabatic and 3 is isothermal.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
