Advertisements
Advertisements
प्रश्न
Amrita electrolysed distilled water using the set-up shown in figure 1. She was expecting two gases to be evolved at the anode and cathode respectively.
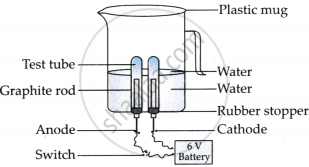
Figure 1
Suddenly, she realised that the bulb in the circuit did not glow when she used distilled water (figure 2).
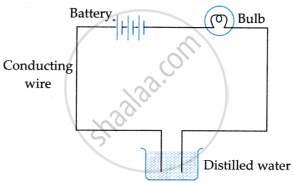
Figure 2
After this realization, she added a substance to the distilled water for electrolysis to take place.
Answer the following questions based on the information given above:
- Which gas was she expecting to be formed at the anode and which one at the cathode respectively?
- Why did the bulb not glow when Amrita passed electricity through distilled water?
- Which substance was added by Amrita to distilled water to get the expected result?
लघु उत्तर
Advertisements
उत्तर
- She was expecting oxygen gas to be formed at the anode and hydrogen at the cathode.
- Distilled water is a poor conductor of electricity.
- Amrita added a few drops of a strong electrolyte like acid H2SO4, or HCl to the distilled water to increase its ion content, making the water conductive and allowing electrolysis to take place.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
