Advertisements
Advertisements
प्रश्न
A light green coloured solution of sulphate salt of metal ‘P’ is taken in a beaker, a rod of another metal ‘Q’ is put in this solution as shown the following figures:
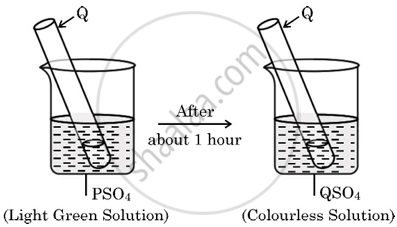
Identify the metals ‘P’ and ‘Q’ and write its chemical equation for the reaction that occurs. State the conclusion of this reaction in terms of reactivity series of metals.
रासायनिक समीकरणे/रचना
लघु उत्तर
Advertisements
उत्तर
Meatal P is iron (Fe).
Metal Q is Zinc (Zn).
The chemical reaction is given as:
\[\ce{Zn + FeSO4 -> \underset{Zinc Sulphate}{ZnSO4} + Fe}\]
In this displacement process, zinc (Zn) replaces iron (Fe) in iron sulphate (FeSO4) due to its higher reactivity. Zinc sulphate (ZnSO4) is formed as a colorless solution, whereas iron (Fe) deposits on the zinc rod.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
