Advertisements
Advertisements
प्रश्न
A certain nucleus has a mass number 20 and atomic number 9. Find the number of neutrons and protons present in it.
Advertisements
उत्तर
(i) Number of neutrons = Mass number – Atomic number
= 20 – 9 = 11
(ii) Number of protons = Atomic number = 9.
संबंधित प्रश्न
Why do Free electrons not leave the metal surface on their own
What do you mean by Atomic number
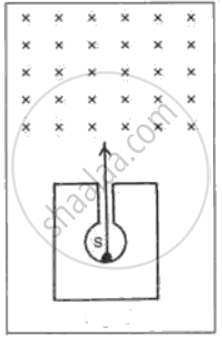
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
What is nucleus of an atom? Compare its size with that of the atom. Name its constitutents. How is the number of these constituents determined by the atomic number its atomic model.
A radioactive substance is oxidized. What changes would you expect to take place in the nature of radioactivity? Explain your answer.
Define bound electrons.
What are cathode rays? Why are they called ‘cathode rays’? Name a material which exhibits fluorescence when cathode rays fall on it.
State one use of radio-isotopes.
The mean lives of a radioactive substance are 1620 and 405 years for β-emission and β-emission respectively. The time after which three fourth of a sample will decay if it is decaying both by β-emission and β-emission simultaneously will be ______ years.
(Take ln 2 = 0.693)
