Advertisements
Advertisements
प्रश्न
A band gap is the distance between the valence band of electrons and the conduction band. Essentially, the band gap represents the minimum energy that is required to excite an electron up to a state in the conduction band where it can participate in conduction. The lower energy level is the valence band, and thus, if a gap exists between this level and the higher energy conduction band, energy must be input for electrons to become free.
An LED is made of a p-type semiconductor material (which has a higher concentration of “holes” or positive charge carriers) and an n-type semiconductor material (which has a higher concentration of electrons or negative charge carriers). This recombination process releases energy in the form of light and heat. The specific wavelength (and therefore the colour) of the emitted light depends on the energy band gap of the semiconductor material used.
The I-V characteristic of the LED bulb is given below.
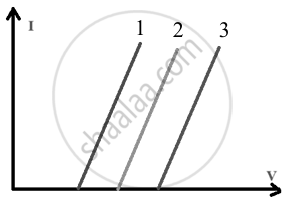
- Identify the wavelength that has:
- The maximum energy gap.
- The minimum energy gap.
- E is the energy of the incident photon and Eg is the energy gap, which is produced across the depletion layer. What will happen in the following cases:
- E > Eg
- E = Eg
- E < Eg
Advertisements
उत्तर
i. In an LED, the energy band gap Eg is related to the wavelength λ of emitted light by:
`E_g ∝ 1/lambda`
Also, the turn-on (threshold) voltage of an LED is directly related to its energy band gap.
a. Curve corresponding to the maximum energy gap is curve 3.
Curve 3 shows the highest threshold (turn-on) voltage; hence, it corresponds to the largest band gap.
b. Curve corresponding to the minimum energy gap is curve 1.
Curve 1 shows the lowest threshold (turn-on) voltage; hence, it corresponds to the smallest band gap.
ii.
- The photon is absorbed and an electron is excited to the conduction band, producing an electron-hole pair. Excess energy may be released as heat.
- The photon is just absorbed and an electron-hole pair is created (threshold condition).
- The photon is not absorbed; hence, no excitation occurs (no electron-hole pair is produced).
