Please select a subject first
Advertisements
Advertisements
Explain the different types of fleshy fruit with suitable example.
Concept: undefined >> undefined
What is the role of national gardens in conserving biodiversity – discuss.
Concept: undefined >> undefined
Advertisements
An example of feedback inhibition is ______.
Concept: undefined >> undefined
Given below is the diagrammatic representation of one of the categories of small molecular weight organic compounds in the living tissues. Identify the category shown & one blank component “ X” in it.
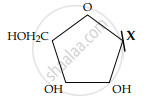
Concept: undefined >> undefined
Distinguish between Nitrogen base and a base found in inorganic chemistry.
Concept: undefined >> undefined
Write the characteristic feature of DNA.
Concept: undefined >> undefined
Explain the structure and function of different types of RNA.
Concept: undefined >> undefined
Give a brief account on Programmed Cell Death (PCD).
Concept: undefined >> undefined
Mention the pigments and storage food of Dinophyceae.
Concept: undefined >> undefined
Name the flagellation found in the zoospore of Oedogonium
Concept: undefined >> undefined
Select the mismatch pair
Concept: undefined >> undefined
Explain the mechanism of biotic stress.
Concept: undefined >> undefined
Where will you place the plants which contain two cotyledons with cup shaped thalamus?
Concept: undefined >> undefined
How do molecular markers work to unlock the evolutionary history of organisms?
Concept: undefined >> undefined
Which of the following is a flowering plant with nodules containing filamentous nitrogen fixing micro - organisms?
Concept: undefined >> undefined
Flowers are zygomorphic in ______.
Concept: undefined >> undefined
Give the floral characters of Clitoria ternatea.
Concept: undefined >> undefined
How will you distinguish Solanaceae members from Liliaceae members?
Concept: undefined >> undefined
If Kb and Kf for a reversible reaction are 0.8 × 10-5 and 1.6 × 10-4 respectively, the value of the equilibrium constant is,
Concept: undefined >> undefined
At a given temperature and pressure, the equilibrium constant values for the equilibria
\[\ce{3A2 + B2 + 2Cr <=>[K1] 2A3BC}\] and
\[\ce{A3BC <=> 3/2 A2 + 1/2 B2 + C}\]
The relation between K1 and K2 is
Concept: undefined >> undefined
