Please select a subject first
Advertisements
Advertisements
Production of iron in a blast furnace follows the following equation:
\[\ce{Fe3O4 (s) + 4CO (g) -> 3Fe (l) + 4CO2 (g)}\]
When 4.640 kg of Fe3O4 and 2.520 kg of CO are allowed to react then the amount of iron (in g) produced is:
[Given: Molar Atomic mass (g mol−1): Fe = 56
Molar Atomic mass (g mol−1): O = 16
Molar Atomic mass (g mol−1): C = 12]
Concept: undefined >> undefined
Sugar moiety in DNA and RNA molecules respectively are ______.
Concept: undefined >> undefined
Advertisements
Given below are two statements:
Statement I: H2O2 can act as both oxidising and reducing agent in basic medium.
Statement II: In the hydrogen economy, the energy is transmitted in the form of dihydrogen.
In the light of the above statements, choose the correct answer from the options given below:
Concept: undefined >> undefined
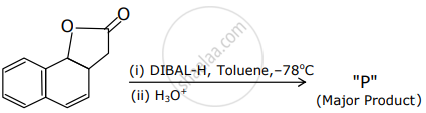
The product "P" in the above reaction is:
Concept: undefined >> undefined
The increasing order of the following compounds towards HCN addition is:
| (i) |  |
| (ii) |  |
| (iii) |  |
| (iv) |  |
Concept: undefined >> undefined
The Born-Haber cycle for KCI is evaluated with the following data:
`Delta "H"_"f"^"o"` for KCl = - 436.7 kJ mol-1;
`Delta "H"_"f"^"o"` for K = 89.2 kJ mol-1
`Delta "H"_"ionization"^"o"` for K = 419.0 kJ mol-1 `Delta "H"_"ionization"^"o"` for Cl(g) = - 348.6 kJ mol-1
`Delta "H"_"bond"^"o"` for Cl2 = 243.0 kJ mol-1
The magnitude of lattice enthalpy of KCl in kJ mol-1 is ______. (Nearest integer)
Concept: undefined >> undefined
AB3 is an interhalogen T-shaped molecule. The number of lone pairs of electrons on A is ______. (Integer answer)
Concept: undefined >> undefined
Identify 'Z' in the reaction given below:
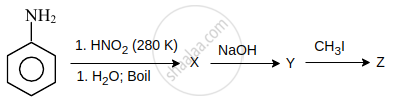
Concept: undefined >> undefined
The free energy change is ______ kJ mol-1 when 1 mole of NaCl is dissolved in water at 25°C. Lattice energy of NaCl = 777.8 kJ mol-1; ΔS for dissolution = 0.043 kJ mol-1; and hydration energy of NaCl = - 774.1 kJ mol-1.
Concept: undefined >> undefined
In the following reaction
\[\ce{Carbonyl compound + MeOH <=>[HCl] acetal}\]
Rate of the reaction is the highest for ______.
Concept: undefined >> undefined
The enthalpy of neutralisation of NH4OH with HCl is −51.46 kJ/mol−1 and the enthalpy of neutralisation of NaOH with HCl is −55.90 kJ/mol−1. The enthalpy of ionisation of NH4OH is ______.
Concept: undefined >> undefined
An aqueous KCl solution of density 1.20 g mL-1 has a molality of 3.30 mol kg-1. The molarity of the solution in mol L-1 is ______. (Nearest integer)
Concept: undefined >> undefined
Which one of the following is formed (mainly) when red phosphorus is heated in a sealed tube at 803 K?
Concept: undefined >> undefined
With increase of temperature, which of these changes?
Concept: undefined >> undefined
What quantity (in mL) of a 45% acid solution of a monoprotic strong acid must be mixed with a 20% solution of the same acid to produce 800 mL of a 29.875% acid solution?
Concept: undefined >> undefined
250 g solution of D-glucose in water contains 10.8% of carbon by weight. The molality of the solution is nearest to ______.
(Given: Atomic weights are H, 1u; C, 12u; O, 16u)
Concept: undefined >> undefined
Bonding in which of the following diatomic molecule(s) become(s) stronger, on the basis of MO Theory, by removal of an electron?
(A) NO
(B) N2
(C) O2
(D) C2
(E) B2
Choose the most appropriate answer from the options given below:
Concept: undefined >> undefined
Given below are the critical temperatures of some of the gases:
| Gas | Critical temperature (K) |
| He | 5.2 |
| CH4 | 190 |
| CO2 | 304.2 |
| NH3 | 405.5 |
The gas showing the least adsorption on a definite amount of charcoal is:
Concept: undefined >> undefined
Lattice enthalpy and enthalpy of the solution of NaCl are 788 kJ mol−1 and 4 kJ mol−1, respectively. The hydration enthalpy of NaCl is ______.
Concept: undefined >> undefined

Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
Concept: undefined >> undefined
