Advertisements
Advertisements
According to IUPAC nomenclatures, sodium nitroprusside is named as
Concept: undefined >> undefined
Which of the following ligands can exhibit linkage isomerism?
Concept: undefined >> undefined
Advertisements
Which alkyl halide has maximum density
Concept: undefined >> undefined
Aldehyde and ketones can de colourize by
Concept: undefined >> undefined
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3C(C2H5 )2CH2Br
Concept: undefined >> undefined
Which one of the following ions is coloured?
Concept: undefined >> undefined
The correct IUPAC name for the given molecule should be
\[\begin{array}{cc}
\ce{H3C - \overset{H}{C} - \overset{H2}{C} - \overset{H}{C} - OH}\\
\phantom{.}|\phantom{.........}|\phantom{}\\
\phantom{...}\ce{CH3}\phantom{......}\ce{CH3}\phantom{}
\end{array}\]
Concept: undefined >> undefined
The element with atomic number 53 belongs to
Concept: undefined >> undefined
Match List-I with List-II:
| List-I | List-II |
| A. ppm | I. molL-¹ |
| B. Molarity | II. No units |
| C. Molality | III. Independent of temperature |
| D. Mole fraction | IV. Very dilute solutions |
Choose the correct answer from the options given below:
Concept: undefined >> undefined
A metallic ion 'M' reacts with chloride ion to form white precipitate which is readily soluble in aqueous ammonia. Identify 'M'?
Concept: undefined >> undefined
The concentration of water molecules in pure water at 298 K is?
Concept: undefined >> undefined
Which of the following I.U.P.A.C. name for (CH3)2CH – CH2 – CH2Br is correct?
Concept: undefined >> undefined
The correct IUPAC name for Na[PdBrCl(NO2)(NH3)] is:
Concept: undefined >> undefined
The element with atomic number 46 belongs to
Concept: undefined >> undefined
 is
is
Concept: undefined >> undefined
Arrange the following compounds in order of decreasing acidity:
| (I) |  |
| (II) | 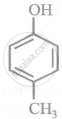 |
| (III) | 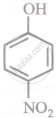 |
| (IV) |  |
Concept: undefined >> undefined
Carbon percentage (by weight) in crude petroleum may be about
Concept: undefined >> undefined
Which of the following is non-metallic?
Concept: undefined >> undefined
The correct IUPAC name of the following compound is:
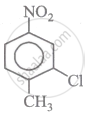
Concept: undefined >> undefined
What is the ratio of mass of an electron to the mass of a proton?
Concept: undefined >> undefined
