Advertisements
Advertisements
Identify the reactant and write the balanced equation for the following:
Nitric acid reacts with compound Q to give a salt Ca(NO3)2, water and carbon dioxide.
Concept: undefined >> undefined
Write balanced equation for a coloured metallic oxide which dissolves in alkalis to yield colourless solutions.
Concept: undefined >> undefined
Advertisements
Write balanced equations for a coloured metallic oxide which dissolves in alkalis to yield colourless solutions.
Concept: undefined >> undefined
In covalent compounds, the bond is formed due to ______ of electrons.
Concept: undefined >> undefined
Explain dipole (polar) molecule by taking hydrogen chloride as an example.
Concept: undefined >> undefined
For each of the salt: A, B, C and D, suggest a suitable method of its preparation.
C is a soluble salt of copper.
Concept: undefined >> undefined
Choosing only substances from the list given in the box below, write equations for the reactions which you would use in the laboratory to obtain: Copper sulphate.
| Dilute sulphuric acid | Copper | Copper carbonate |
| Iron | Sodium carbonate | |
| Sodium | ||
| Zinc |
Concept: undefined >> undefined
Choosing only substances from the list given in the box below, write equations for the reactions which you would use in the laboratory to obtain: lron(II) sulphate
| Dilute sulphuric acid | Copper | Copper carbonate |
| Iron | Sodium carbonate | |
| Sodium | ||
| Zinc |
Concept: undefined >> undefined
Choosing only substances from the list given in the box below, write equations for the reactions which you would use in the laboratory to obtain: Zinc carbonate.
| Dilute sulphuric acid | Copper | Copper carbonate |
| Iron | Sodium carbonate | |
| Sodium | ||
| Zinc |
Concept: undefined >> undefined
Give equations for the following conversions A to E.
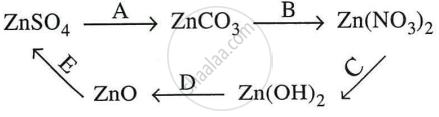
Concept: undefined >> undefined
Give a balanced equation for the reaction:
Silver nitrate solution and sodium chloride solution.
Concept: undefined >> undefined
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
Concept: undefined >> undefined
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
Concept: undefined >> undefined
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Concept: undefined >> undefined
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
Concept: undefined >> undefined
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Concept: undefined >> undefined
What do you understand by atomic size? State its unit?
Concept: undefined >> undefined
Give the trends in atomic size on moving down the group.
Concept: undefined >> undefined
Give the trends in atomic size on moving across the period left to right.
Concept: undefined >> undefined
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
Concept: undefined >> undefined
