Advertisements
Advertisements
Write the IUPAC name of the following complex:
[Pt(NH3)6]Cl4
Concept: Aldehydes and Ketones >> Nomenclature of Aldehydes and Ketones
Using IUPAC norms, write the formula for the following:
Pentaamminenitrito-N-Cobalt (III)
Concept: Aldehydes and Ketones >> Nomenclature of Aldehydes and Ketones
Using IUPAC norms write the formula for the following:
Tetrahydroxidozincate (II)
Concept: Aldehydes and Ketones >> Nomenclature of Aldehydes and Ketones
Predict the reagent for carrying out the following transformations:
Benzoyl chloride to Benzaldehyde
Concept: Aldehydes and Ketones >> Preparation of Aldehydes
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Which of the following does not give aldol condensation reaction?
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Which of the following does not give Cannizzaro reaction?
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Other Reactions
Aldehydes and ketones react with hydroxylamine to form ______.
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
The reagent that can be used to distinguish acetophenone and benzophenone is ______.
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Oxidation
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Do the following conversions in not more than two steps:
CH3COOH to CH3COCH3
Concept: Aldehydes and Ketones >> Structure of the Carbonyl Group
Do the following conversions in not more than two steps:
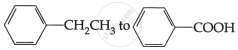
Concept: Aldehydes and Ketones >> Structure of the Carbonyl Group
Assertion (A): Acetic acid but not formic acid, can be halogenated in the presence of red P and Cl2.
Reason (R): Acetic acid is a weaker acid than formic acid.
Concept: Acids >> Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen

Identify A and B:
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
What is IUPAC name of the ketone A, which undergoes iodo form reaction to give \[\ce{CH3CH = C(CH3)COONa}\] and yellow precipitate of \[\ce{CH3}\]?
Concept: Aldehydes and Ketones >> Nomenclature of Aldehydes and Ketones
Assertion (A): Strong oxidising agents oxidise toluene and its derivatives to benzoic acids.
Reason (R): It is possible to stop the oxidation of toluene at the aldehyde stage with suitable reagents.
Select the most appropriate answer from the options given below:
Concept: Aldehydes and Ketones >> Preparation of Aldehydes
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Write the structure of the product formed when acetone reacts with 2, 4 DNP reagent.
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Convert the following:
Benzene to m-nitrobenzaldehyde
Concept: Aldehydes and Ketones >> Chemical Reactions of Aldehydes and Ketones - Other Reactions
