Advertisements
Advertisements
What should be the signs (positive/negative) for \[\ce{E^0_{cell}}\] and ΔG0 for a spontaneous redox reaction occurring under standard conditions?
Concept: Electrochemical Cells
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
| Rahul set up an experiment to find the resistance of aqueous KCl solution for different concentrations at 298 K using a conductivity cell connected to a Wheatstone bridge. He fed the Wheatstone bridge with a.c. power in the audio frequency range 550 to 5000 cycles per second. Once the resistance was calculated from the null point, he also calculated the conductivity K and molar conductivity ∧m and recorded his readings in tabular form. |
| S. No. | Conc. (M) |
k S cm−1 | ∧m S cm2 mol−1 |
| 1. | 1.00 | 111.3 × 10−3 | 111.3 |
| 2. | 0.10 | 12.9 × 10−3 | 129.0 |
| 3. | 0.01 | 1.41 × 10−3 | 141.0 |
Answer the following questions:
(a) Why does conductivity decrease with dilution? (1)
(b) If `∧_"m"^0` of KCl is 150.0 S cm2 mol−1, calculate the degree of dissociation of 0.01 M KCI. (1)
(c) If Rahul had used HCl instead of KCl then would you expect the ∧m values to be more or less than those per KCl for a given concentration? Justify. (2)
OR
(c) Amit a classmate of Rahul repeated the same experiment with CH3COOH solution instead of KCl solution. Give one point that would be similar and one that would be different in his observations as compared to Rahul. (2)
Concept: Conductance of Electrolytic Solutions >> Variation of Conductivity and Molar Conductivity with Concentration
Define “zero order reaction”.
Concept: Factors Influencing Rate of a Reaction
Write two factors that affect the rate of reaction.
Concept: Factors Influencing Rate of a Reaction
For a reaction : 
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
Concept: Factors Influencing Rate of a Reaction
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
Concept: Temperature Dependence of the Rate of a Reaction
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
Concept: Factors Influencing Rate of a Reaction
A first order reaction takes 30 minutes for 50% completion. Calculate the time required for 90% completion of this reaction.
(log 2 = 0.3010)
Concept: Half Life Period of a Reaction
The decomposition of NH3 on platinum surface is zero order reaction. What are the rates of production of N2 and H2 if k = 2.5 × 10−4 mol−1 L s−1?
Concept: Zero Order Reactions
Following data are obtained for reaction :
N2O5 → 2NO2 + 1/2O2
| t/s | 0 | 300 | 600 |
| [N2O5]/mol L–1 | 1.6 × 10-2 | 0.8 × 10–2 | 0.4 × 10–2 |
1) Show that it follows first order reaction.
2) Calculate the half-life.
(Given log 2 = 0.3010, log 4 = 0.6021)
Concept: First Order Reactions
Write the expression of integrated rate equation for zero order reaction.
Concept: Zero Order Reactions
Observe the graph shown in figure and answer the following questions:
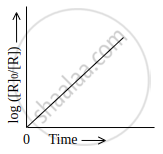
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
Concept: First Order Reactions
Read the following passage and answer the questions that follow:
|
The rate of reaction is concerned with decrease in the concentration of reactants or increase in the concentration of products per unit of time. It can be expressed as instantaneous rate at a particular instant of time and average rate over a large interval of time. A number of factors such as temperature, concentration of reactants, catalyst affect the rate of reaction. Mathematical representation of rate of a reaction is given by rate law: Rate = k[A]x [B]y x and y indicate how sensitive the rate is to change in concentration of A and B. Sum of x + y gives the overall order of a reaction. |
- What is the effect of temperature on the rate constant of a reason? [1]
- For a reaction \[\ce{A + B → Product}\], the rate law is given by, Rate = k[A]2 [B]1/2. What is the order of the reaction? [1]
- How order and molecularity are different for complex reactions? [1]
- A first-order reaction has a rate constant 2 × 10–3 s–1. How long will 6 g of this reactant take to reduce to 2 g? [2]
OR
The half-life for radioactive decay of 14C is 6930 years. An archaeological artifact containing wood had only 75% of the 14C found in a living tree. Find the age of the sample.
[log 4 = 0.6021, log 3 = 0.4771, log 2 = 0.3010, log 10 = 1] [2]
Concept: Factors Influencing Rate of a Reaction
The following experimental rate data were obtained for a reaction carried out at 25°C:
\[\ce{A_{(g)} + B_{(g)} -> C_{(g)} + A_{(g)}}\]
| Initial [A(g)]/mol dm−3 | Initial [B(g)]/mol dm−3 | Initial rate/mol dm−3s−1 |
| 3.0 × 10−2 | 2.0 × 10−2 | 1.89 × 10−4 |
| 3.0 × 10−2 | 4.0 × 10−2 | 1.89 × 10−4 |
| 6.0 × 10−2 | 4.0 × 10−2 | 7.56 × 10−4 |
What are the orders with respect to A(g) and B(g)?
Concept: Zero Order Reactions
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
Concept: Temperature Dependence of the Rate of a Reaction
Account for the following:
Cr2+ is a strong reducing agent.
Concept: Physical Properties of the Transition Elements (d-block)
When chromite ore FeCr2O4 is fused with NaOH in presence of air, a yellow-coloured compound (A) is obtained, which on acidification with dilute sulphuric acid gives a compound (B). Compound (B) on reaction with KCl forms an orange coloured crystalline compound (C).
(i) Write the formulae of the compounds (A), (B) and C.
(ii) Write one use of compound (C).
Concept: Important Compounds of Transition Elements
Complete the following chemical equation
8MnO4- + 3S2O32- + H2O →
Concept: Important Compounds of Transition Elements
What are the transition elements? Write two characteristics of the transition elements.
Concept: Physical Properties of the Transition Elements (d-block)
How would you account for the following:
Actinoid contraction is greater than lanthanoid contraction.
Concept: F-block Elements >> The Actinoids
