Definitions [22]
The substance adsorbed on the surface of another substance is called as adsorbate or absorbed phase.
A substance which adsorbs another substance on its surface is called as adsorbent.
Define adsorption.
Adsorption is the phenomenon of accumulation of a higher concentration of one substance on the surface of another (in bulk) due to unbalanced/unsatisfied attractive forces on the surface.
The phenomenon of accumulation of substance at the surface of another molecular species rather than in its bulk is called adsorption. e.g. Adsorption of gases like nitrogen, carbon dioxide by activated charcoal.
When both adsorption and absorption occur simultaneously, it is known as sorption.
The process of removal of an adsorbed substance from the surface of adsorbent is known as desorption.
The relationship between the extent of adsorption (x/m) and the pressure of gas at constant temperature is called an adsorption isotherm.
Define the term Inhibition.
The phenomenon in which the rate of a chemical reaction is reduced by an inhibitor is called inhibition.
A catalyst is a substance that alters the rate of a chemical reaction without being consumed. The phenomenon is called catalysis.
Define the term Catalysis.
The phenomenon of increasing the rate of a chemical reaction with the help of a catalyst is known as catalysis.
Define the term Electrophoresis.
The movement of colloidal particles under an applied electric potential is called electrophoresis.
A substance is said to be in colloidal state when one substance of heterogeneous system is dispersed (dispersed phase) as very fine particles in another substance (dispersion medium).
Define the following term:
Multimolecular colloids
Multimolecular colloids are species having size in the colloidal range (diameter < 1 nm) that are formed by the aggregation of large number of atoms or small molecules of substances when dissolved in the dispersion medium.
The process used for reducing the amount of impurities to a requisite minimum is called as purification of colloidal solution.
Dialysis process is used for removing a dissolved substance from a colloidal solution by diffusion through a suitable membrane.
Define the following term:
Multimolecular colloids
Multimolecular colloids are species having size in the colloidal range (diameter < 1 nm) that are formed by the aggregation of large number of atoms or small molecules of substances when dissolved in the dispersion medium.
The process used for reducing the amount of impurities to a requisite minimum is called as purification of colloidal solution.
Dialysis process is used for removing a dissolved substance from a colloidal solution by diffusion through a suitable membrane.
Define the following term:
Zeta potential
The potential difference between a colloidal particle's fixed layer (primary layer) and diffused layer (secondary layer) is known as the zeta potential. It's also referred to as electrokinetic potential.
Define coagulation.
It is a process of aggregating together the colloidal particles so as to change them into large-sized particles, which ultimately settle as a precipitate.
Define the following term:
Coagulation
Coagulation is the process by which colloidal particles aggregate and settle as a precipitate. It is brought about by the addition of electrolyte.
Emulsions are colloidal solutions of two immiscible liquids in which one liquid acts as the dispersed phase and the other as the dispersion medium.
Key Points
| Feature | Physisorption | Chemisorption |
|---|---|---|
| Force | Van der Waals' | Covalent / ionic bond |
| Specificity | Non-specific | Highly specific |
| Heat of adsorption | 20–40 kJ mol⁻¹ | 40–200 kJ mol⁻¹ |
| Temperature | Low temp favoured | High temp favoured |
| Reversibility | Reversible | Irreversible |
| Layers | Multilayer | Monolayer |
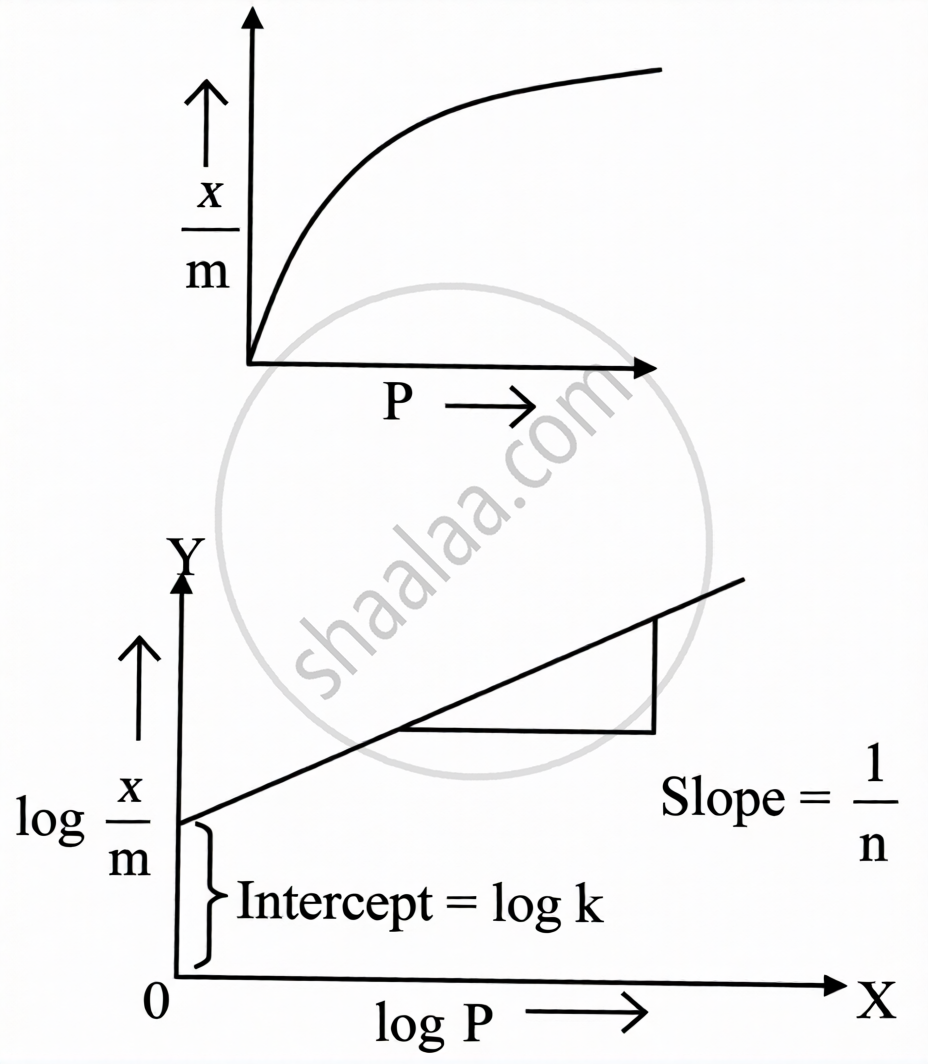
Freundlich Adsorption Isotherm:
Empirical equation: \[\frac{x}{m}=kP^{1/n}\quad(n>1)\]
where:
- x = mass of the gas adsorbed
- m = mass of the adsorbent
- `x/m` = mass of gas adsorbed per unit mass of adsorbent
- P = equilibrium pressure
- k and n = constants depending on the nature of the adsorbate, adsorbent, and temperature
For solutions: \[\frac{x}{m}=kC^{1/n}\] where C = concentration of the solution
Logarithmic (linear) form: \[\log\frac{x}{m}=\log k+\frac{1}{n}\log P\]
In the log x/m vs log P graph:
- Slope = `1/n` (value between 0 and 1)
- Y-intercept = log k
- When `1/n` = 0: adsorption is independent of pressure
- When `1/n` = 1: adsorption is directly proportional to pressure
| Application | How Adsorption Helps |
|---|---|
| Adsorption indicators | Dyes like eosin and fluorescein act as indicators through adsorption |
| Separation of inert gases | Inert gases can be separated on coconut charcoal (different adsorption at different temperatures) |
| Froth flotation process | Hydrophobic pine oil preferentially adsorbs sulphide ore particles, allowing separation from gangue |
| Chromatographic analysis | Powdered adsorbents like silica or alumina gel are used to separate mixtures |
| Heterogeneous catalysis | Contact process (SO₂ → SO₃), hydrogenation of oils |
| Gas masks | Activated charcoal or mixture of adsorbents to avoid inhaling poisonous gases |
| Control of humidity | Silica and alumina gels used to adsorb moisture |
| Production of high vacuum | Coconut charcoal adsorbs traces of air or moisture to create high vacuum |
- Nature of adsorbent — Substances with large surface area per unit mass (porous solids like silica gel and charcoal) are more effective adsorbents
- Surface area — Greater the surface area of the adsorbent, greater is the extent of adsorption
- Temperature — Adsorption is exothermic, so it decreases with rise in temperature (and vice versa)
- Pressure of the gas — At constant temperature, extent of gas adsorption increases with increase in pressure
- Nature of adsorbate (gas) — Gases that are more easily liquefiable (e.g., SO₂, Cl₂, NH₃) get adsorbed to a larger extent compared to gases like N₂, O₂, H₂
| Type | Description |
|---|---|
| Homogeneous |
Reactants and catalyst are in the same phase \[\ce{\underset{}{2SO2(g) + O2(g)} ->[NO(g)][]\underset{}{2SO3(g)}}\] |
| Heterogeneous |
Reactants and catalyst are in different phases \[\ce{\underset{}{N2(g) + 3H2(g)} ->[][Fe(s)]\underset{}{2NH3(g)}}\] |
| On the basis of the mechanism of catalysis: |
|
| Positive catalysis | Catalyst enhances the rate of reaction |
| Negative catalysis | Catalyst retards the rate of reaction |
| Auto-catalysis | One of the products acts as catalyst |
| Induced catalysis | One reaction induces the rate of another reaction |
The reaction occurs on the surface of the solid catalyst. Steps involved:
- Diffusion — Reactant molecules diffuse towards the surface of the catalyst
- Adsorption — Reactant molecules adsorb onto the surface of the catalyst
- Intermediate formation — Chemical reaction on the catalyst surface proceeds via intermediate formation
- Desorption — Products are desorbed (released) from the catalyst surface
- Product formation — Products diffuse away from the surface
Heterogeneous catalytic reactions are also called surface reactions. Heterogeneous catalysts are used in automobile catalytic converters.
(i) Based on Physical State of Dispersed Phase and Dispersion Medium
| Dispersed Phase | Dispersion Medium | Type | Examples |
|---|---|---|---|
| Solid | Solid | Solid sol | Coloured glass, gem stones, porcelain, paper |
| Solid | Liquid | Sol / Gel | Paints, starch solution, gold sol, muddy water |
| Solid | Gas | Aerosol | Smoke, dust |
| Liquid | Solid | Gel | Cheese, butter, jellies |
| Liquid | Liquid | Emulsion | Milk, hair cream |
| Liquid | Gas | Aerosol | Fog, mist, cloud, insecticide sprays |
| Gas | Solid | Solid sol | Pumice stone, foam rubber, plaster |
| Gas | Liquid | Foam | Froth, whipped cream, soap lather |
(ii) Based on Interaction / Affinity of Phases
| Feature | Lyophilic Sols | Lyophobic Sols |
|---|---|---|
| Meaning | Liquid-loving (lyo = solvent, philic = loving) | Liquid-hating (phobic = hating) |
| Preparation | Formed easily by direct mixing | Formed only by special methods |
| Stability | Self-stabilized; reversible | Unstable; require stabilizers; irreversible |
| Electrolyte effect | Large amount of electrolyte causes coagulation | Small amount of electrolyte causes coagulation |
| Viscosity | Much higher than dispersion medium | Nearly same as dispersion medium |
| Surface tension | Lower than dispersion medium | Nearly same as dispersion medium |
| Examples | Starch, gelatin, proteins, gum | Au sol, As₂S₃, Fe(OH)₃ |
(iii) Based on Molecular Size
| Type | Description | Examples |
|---|---|---|
| Multimolecular colloids | Aggregates of atoms or small molecules; size < 10³ pm | Gold sol, sulphur molecules |
| Macromolecular colloids | Individual molecules are large enough to be colloidal dimensions | Starch, cellulose, proteins, polythene, nylon, plastics |
| Associated colloids / Micelles | Normal electrolytes at low concentration; form colloidal aggregates (micelles) at high concentration | Soaps, detergents |
| Property Type | Property | Description |
|---|---|---|
| General Property | Nature | Heterogeneous system; particles pass slowly through membranes |
| Kinetic Property | Brownian Motion | Continuous random zig-zag motion of colloidal particles |
| Optical Property | Tyndall Effect | Scattering of light makes path of beam visible |
|
Colour |
Depends on wavelength of scattered light | |
| Electrical Property | Charge | Colloidal particles carry electric charge |
| Electrophoresis | Movement of particles towards oppositely charged electrode | |
| Electro-osmosis | Movement of dispersion medium under electric field | |
| Mechanical Property | Brownian Motion | Particles remain in constant motion |
| Stability Property | Coagulation | Precipitation of colloidal particles |
| Flocculation Value | ∝ 1 / Coagulating power |
1. Chemical Methods
-
Oxidation: \[\mathrm{SO}_2+2\mathrm{H}_2\mathrm{S}\xrightarrow{\mathrm{Oxidation}}3\mathrm{S}\downarrow+2\mathrm{H}_2\mathrm{O}\] (sulphur sol)
-
Reduction: \[2\mathrm{AuCl}_{3}+3\mathrm{HCHO}+3\mathrm{H}_{2}\mathrm{O}\xrightarrow{\mathrm{Reduction}}2\mathrm{Au}\downarrow+3\mathrm{HCOOH}+6\mathrm{HCl}\] (gold sol)
- Hydrolysis: \[\mathrm{FeCl}_3+3\mathrm{H}_2\mathrm{O}\xrightarrow{\text{Hydrolysis}}\mathrm{Fe(OH)}_3\downarrow+3\mathrm{HCl}\] (iron hydroxide sol)
2. Electrical Disintegration — Bredig's Arc Method
- An electric arc is struck between electrodes of the metal immersed in the dispersion medium
- The intense heat produces metal vapour that condenses to form colloidal particles
3. Peptization
-
A precipitate is converted into colloidal sol by shaking it with the dispersion medium in the presence of a small amount of electrolyte (the peptizing agent)
Emulsions show all properties of sols. Their particle size is 1000 Å to 10,000 Å; they scatter light (Tyndall effect).
Types of Emulsions:
| Feature | Oil in Water (O/W) | Water in Oil (W/O) |
|---|---|---|
| Dispersed phase | Oil | Water |
| Dispersion medium | Water | Oil |
| Continuous phase | Water | Oil |
| Miscibility | Addition of water mixes with it | Addition of oil mixes with it |
| Electrolyte effect | Small amount makes it conducting | No effect on conducting power |
| Emulsifiers used | Water-soluble alkali metal soaps, metal sulphates | Water-insoluble soaps (Zn, Al, Fe), alkaline earth metal soaps |
| Examples | Milk, vanishing cream | Butter, cream, cod liver oil |
Concepts [26]
- Surface Chemistry
- Introduction of Adsorption
- Distinction Between Adsorption and Absorption
- Adsorption
- Mechanism of Adsorption
- Types of Adsorption
- Adsorption Isotherms (Freundlich and Langmuir Adsorption Isotherm)
- Applications of Adsorption
- Factors Affecting Adsorption of Gases on Solids
- Adsorption from Solution Phase
- Catalysis
- Homogeneous and Heterogeneous Catalysis
- Shape-selective Catalysis by Zeolites
- Enzyme Catalysis
- Catalysts in Industry
- Adsorption Theory of Heterogeneous Catalysis
- Catalytic Activity and Selectivity
- Colloids
- Classification of Colloids
- Purification of Colloidal Solution
- Properties of Colloidal Dispersions
- Preparation of Colloids
- Purification of Colloidal Solution
- Properties of Colloidal Solutions
- Colloids Around Us
- Emulsions
