Advertisements
Advertisements
प्रश्न
Write the structural difference between white phosphorus and red phosphorus.
Advertisements
उत्तर
Structures
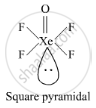
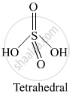
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
Write the structural difference between white phosphorus and red phosphorus.
Structures

