Advertisements
Advertisements
प्रश्न
Write the electronic configuration of two elements X and Y whose atomic numbers are 20 and 17 respectively. Write the molecular formula of the compound formed when element X reacts with element Y. Draw the electron-dot structure of the product and also state the nature of the bond formed between both the elements.
Advertisements
उत्तर
Atomic number of element X = 20
Electronic configuration of element X = 2, 8, 8, 2
Atomic number of element Y = 17
Electronic configuration of element Y = 2, 8, 7
When X reacts with Y, it looses the two valence electrons (valency 2). These two valence electrons are accepted by two Y atoms (valency 1). Hence, the molecular formula of the compound formed between X and Y is XY2 .
Electron-dot structure
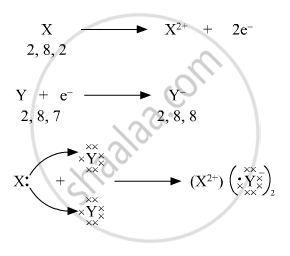
Ionic bond is formed between the elements X and Y in compound XY2.
