Advertisements
Advertisements
प्रश्न
Write down the formulae of sodium sulphide.
लघु उत्तरीय
Advertisements
उत्तर
| Ions present | Formula |
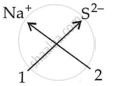 |
Na2S |
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 3: Atoms and Molecules - Intext question [पृष्ठ ३४]
APPEARS IN
संबंधित प्रश्न
Correct the following statement:
A molecular formula represents an element.
XCl2 is the chloride of a metal X. State the formula of the sulphate and the hydroxide of the metal X.
Write the formula of the following compound:
Potassium sulphite
Write the formula of the following compound:
Potassium permanganate
Write the formula of the following compound:
Potassium hydroxide
Write the formula of the following compound:
Sodium nitrate
Write the formula of the following compound:
Calcium chloride
Write the formula of the following compound:
Calcium sulphite
Write the formula of the following compound:
Tetra amine copper [II] sulphate
Write the formula of the following compound:
Iron [II] sulphate
