Advertisements
Advertisements
प्रश्न
Write balanced chemical equations to show The behavior of H2SO4 as an acid when it reacts with Magnesium
Advertisements
उत्तर
Mg + H2SO4  MgSO4 + H2↑
MgSO4 + H2↑
APPEARS IN
संबंधित प्रश्न
Complete the following by selecting the correct option from the choices given:
The metal which does not react with water or dilute H2SO4 but reacts with concentrated H2SO4 is _________. (Al/Cu/Zn/Fe)
Select from the list the gas that matches the description given in case
[ammonia, ethane, hydrogen chloride, hydrogen sulfide, ethyne]
This gas has a characteristic rotten egg smell.
Name the gas that is produced in the given cases :
An action of dilute hydrochloride acid on sodium sulphide.
Give a chemical test to distinguish between the given pairs of chemicals:
Sodium chloride solution and Sodium nitrate solution
Write an equation for the reaction of aqueous hydrochloric acid on lead nitrate solution.
State the use of aqua-regia.
Write a fully balanced of the following case :
Red lead is warmed with concentrated hydrochloric acid.
Write a fully balanced of the following case :
Magnesium metal is treated with dilute hydrochloric acid.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
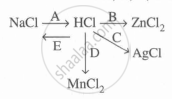
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.