Advertisements
Advertisements
प्रश्न
What is the oxidation state of sulphur in the following?
Sulphurous acid
What is the oxidation state of S in the following oxoacid?
Sulphurous acid
Advertisements
उत्तर
Sulphurous acid (H2SO3)
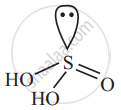
2 × (+1) + (Oxidation state of S) + 3 × (−2) = 0
2 + (Oxidation state of S) − 6 = 0
Hence, the oxidation state of ‘S’ in sulphurous acid (H2SO3) is +4.
संबंधित प्रश्न
Answer the following.
What is the oxidation state of ‘S’ in H2SO4?
Answer the following.
What happens when Cl2 reacts with F2 in equal volume at 437 K.
Answer the following.
What happens when Br2 reacts with excess of F2.
Write a balanced chemical reaction of sulfuric acid with carbon.
What is oxidation state of sulphur in the following?
Peroxymonosulfuric acid
Write three physical properties of sulfuric acid.
Draw the structure of chloric acid.
The formula of chlorous acid is ____________.
Which of the following molecular formula represents Marshall's acid?
How many numbers of P—O—H and P—O—P bonds are present in pyrophosphoric acid respectively?
The following structure represents

In which of the following oxoacids 'Cl' exhibit +5 oxidation state?
The molecular formula of peroxymonosulfuric acid is ____________.
Which of the following structure represents thiosulfuric acid?
The molecular formula of peroxydisulfuric acid is ____________.
Which of the following oxyacids of chlorine does not contain lone pair of electron on chlorine atom?
Which of the following halogen does NOT form perhalic acid?
Which of the following structure represents chloric acid?
Which of the following oxyacid of sulphur contain both S=S and S=O bonds?
What is the oxidation state of chlorine atom in hypochlorous acid?
Which among the following halogens does not form the oxide of type OX2? (X = halogen)
How many lone pair of electrons are present on chlorine atoms in Hypochlorous acid?
Write the structure of HClO4.
Draw the structure of oxyacid of sulphur in which the oxidation state of sulphur is + 4.
Draw the structure of peroxymonosulphuric acid.
