Advertisements
Advertisements
प्रश्न
Using valence bond theory, explain the hybridisation and magnetic behaviour of the following:
K2[NiCl4]
(At. no.: Ni = 28)
स्पष्ट कीजिए
Advertisements
उत्तर
The central metal ion in this complex is Ni2+, which has an atomic number of 28. Its electronic configuration is [Ar] 3d8 4s0. Chloride ions (Cl−) are weak-field ligands, so they do not cause pairing of electrons in the d-orbitals. As a result, the 3d electrons in Ni2+ remain unpaired. For a complex with four ligands and no pairing, the hybridisation involves one 3d, one 4s, and two 4p orbitals, leading to sp3 hybridisation.
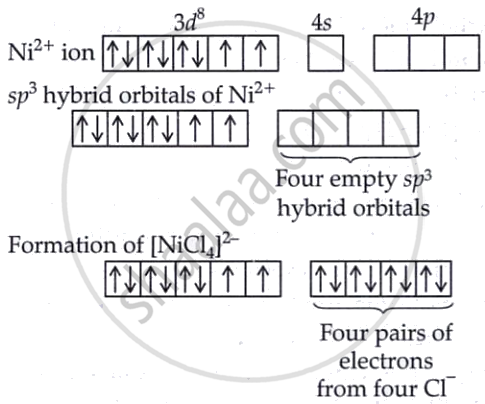
This sp3 hybridisation gives the complex a tetrahedral geometry. Since there are two unpaired electrons in the 3d orbitals, the complex shows paramagnetic behaviour.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
2024-2025 (March) Outside Delhi Set 3
