Advertisements
Advertisements
प्रश्न
The stable carbocation formed in the below reaction is

विकल्प
\[\ce{CH3 CH2 C\overset{⊕}{H}_2}\]
\[\ce{CH3 C\overset{⊕}{H}_2}\]
\[\ce{CH3 - \overset{⊕}{C}H - CH3}\]
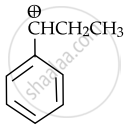
MCQ
Advertisements
उत्तर
\[\ce{CH3 - \overset{⊕}{C}H - CH3}\]
Explanation:
\[\begin{array}{cc}
\ce{CH3CH2CH2Cl + Anhyd. AlCl3}\\
\phantom{.}↓\\
\ce{\underset{(1°)}{CH3CH2CH_2^+} + AlCl_4^⊖}\phantom{..........}\end{array}\]
\[\ce{\underset{\phantom{......}(1°)}{CH3\overset{2}{C}H2}\overset{1}{C}H_2^+ <=>[Rearrangement] \underset{(2°)}{CH3\overset{⊕}{C}HCH3}}\]
After 1, 2 - Hydride shift, 1° carbocation is converted to 2° carbocation.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
