Advertisements
Advertisements
प्रश्न
The following sketch illustrates the process of conversion of Alumina to Aluminium:
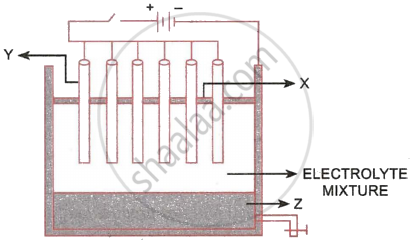
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
संक्षेप में उत्तर
Advertisements
उत्तर
- The electrolytes used are molten alumina, cryolite and fluospar.
- Powdered coke layer (X).
- Hall Heroult process
- Cathode (Z): \[\ce{Al^{3+} + 3e^{-} -> Al}\]
Anode (Y): \[\ce{C + 2O^{2+} -> CO_2 + 4e^{-}}\]
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
