Advertisements
Advertisements
प्रश्न
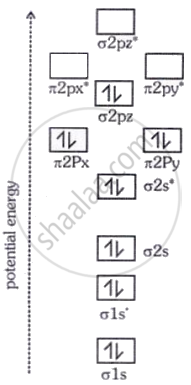
The correct order of energies of molecular orbitals of the N molecule is:
विकल्प
σ1s < σ*1s < σ2s < σ*2s < σ2p < (π2px = π2py) < (π*2px = π*2py) < σ*2pz
σ1s < σ*1s < σ2s < σ*2s < σ2p < σ*2pz < (π2px = π2py) < (π*2px = π*2py)
σ1s < σ*1s < σ2s < σ*2s < (π2px = π2py) < (π*2px = π*2py) < σ2p < σ*2pz
σ1s < σ*1s < σ2s < σ*2s < (π2px = π2py) < σ2p < (π*2px = π*2py) < σ*2pz
MCQ
Advertisements
उत्तर
σ1s < σ*1s < σ2s < σ*2s < (π2px = π2py) < σ2p < (π*2px = π*2py) < σ*2pz
Explanation:
Molecular orbital (energy) diagram/sequence of N2:
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
