Advertisements
Advertisements
प्रश्न
Select the chemical changes from the following list of changes
Preparation of carbon dioxide from calcium carbonate & dilute hydrochloric acid.
Advertisements
उत्तर
Preparation of carbon dioxide from calcium carbonate and dilute hydrochloric acid is a CHEMICAL change as new K substance with new properties is formed, the change is permanent and irreversible.
APPEARS IN
संबंधित प्रश्न
Write true or false for the statement.
Deforestation is a man-made change
Dissolving salt in water is a reversible change.
Tick the most appropriate answer.
Physical changes are
A physical change is temporary. Explain.
Solve this crossword by using the clues that follow-
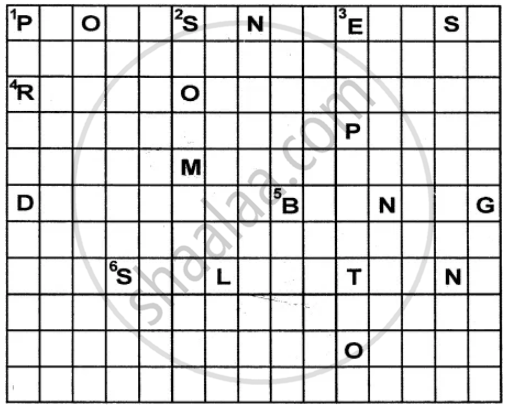
Across
1. Energy in the form of sunlight is absorbed by the green plants in this process.
4. The reddish-brown substance formed over iron in the presence of oxygen and moisture.
5. This change is permanent and irreversible.
6. The process in which a solid directly changes into gaseous state.
Down
1 . It is the change that takes place in case of swinging pendulum of a clock. –
2. Occurrence of this is a non-periodic change as well as a natural change.
3. It is a physical change.
Fill in the blanks with the appropriate words of the statement given below:
Heating a piece of iron is considered a __________ change while heating a piece of coal [carbon] is considered a ___________ change.
In a solution of salt in water, water is the solute
When a woolen yarn is knitted to get a sweater, the change can be classified as ____________.
Analogy:
Melting of ice: Physical change.
Popping of popcorn: ______.
Two drops of dilute sulphuric acid were added to 1 g of copper sulphate powder and then small amount of hot water was added to dissolve it (step I). On cooling, beautiful blue coloured crystals got separated (step II). Step I and Step II are:
