Advertisements
Advertisements
प्रश्न
| Phenol is an aromatic alcohol that is used to prepare many important compounds such as picric acid. Phenol is widely used in household and industrial settings as a cleaner and disinfectant. It is also used as a primary chemical to make plastics. Phenol is less soluble in water as compared to aliphatic alcohol. Some aliphatic alcohols are toxic and can be addictive. |
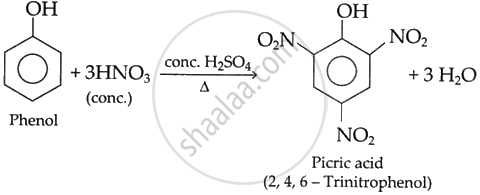
- How is the acid mentioned above prepared from phenol? Write the chemical reaction involved in this preparation.
- ‘Phenol is less soluble in water as compared to aliphatic alcohol.’ Explain.
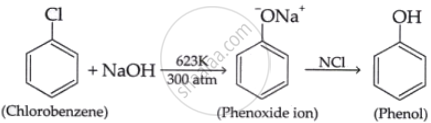
- Write the chemical equation for the preparation of phenol from chlorobenzene.
- An organic compound [A] having molecular formula C4H10O gives positive Lucas test within five minutes at room temperature. Compound [A] upon oxidation with K2Cr2O7/H2SO4 forms compound [B] which does not respond to Tollen’s test.
Identify compounds [A] and [B]. - In the above reaction, compound [B] gets reduced with Zn/Hg and HCl and forms compound [C]. Identify compound [C] and write the balanced reaction for the conversion of compound [B] to compound [C].
Advertisements
उत्तर
i. Phenol can be converted into picric acid (2,4,6-trinitrophenol) by nitration using concentrated HNO3 (Nitric Acid) in the presence of H2SO4 (Sulphuric Acid).
ii. The phenol dissolves in water because it has a hydroxyl (–OH) group that can make hydrogen bonds with water molecules. But phenol also has a big benzene ring that doesn’t like water, which makes it harder to dissolve in water. On the other hand, aliphatic alcohols have fewer nonpolar areas and are more likely to form hydrogen bonds, which makes them easier to dissolve in water. As the benzene ring, the hydrophobic part of phenol, gets bigger, the hydrogen bonds with water get smaller, which makes the phenol less soluble.
iii.
iv. Alcohols are distinguished by their reactivity using the Lucas test. Compound [A] (C4H10O) must be a secondary alcohol because it produces a positive Lucas test in less than five minutes (whereas tertiary alcohols produce an immediate test).
Oxidation with K2Cr2O7/H2SO4 converts secondary alcohols into ketones.
[B] does not respond to Tollen’s test, meaning it is not an aldehyde, confirming that [B] is a ketone.
The possible secondary alcohol with C4H10O is butan-2-ol. Thus,
[A] = Butan-2-ol (CH3 – CHOH – CH2 – CH3)
[B] = Butan-2-one (CH3 – CO – CH2 – CH3, also called Methyl Ethyl Ketone)
\[\ce{\underset{Butan-2-ol}{\underset{[A]}{CH3 - CHOH - CH2 - CH3}} + [O] ->[K2Cr2O7/H2SO4] \underset{Butan-2-one}{\underset{[B]}{CH3 - CO - CH2 - CH3}} + H2O}\]
v. Compound [B] (butan-2-one) undergoes Clemmensen reduction (Zn/Hg + HCl), which converts it into an alkane (butane, C4H10). The carbonyl (–CO) group is completely reduced to –CH2, giving butane (C4H10).
\[\ce{\underset{[B]}{\underset{Butan-2-one}{CH3 - CO - CH2 - CH3}} + 4[H] ->[Zn/Hg*HCl] \underset{[C]}{\underset{Butane}{CH3 - CH2 - CH2 - CH3}} + H2O}\]
