Advertisements
Advertisements
प्रश्न
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is ___________.
विकल्प
propan-2-ol
propan-1-ol
ethoxy ethane
methoxy ethane
Advertisements
उत्तर
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is methoxy ethane.
APPEARS IN
संबंधित प्रश्न
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroxylation using bayers reagent
Complete the following reaction.
\[\ce{C6H5 - OH ->[Zn dust] A ->[CH3Cl][Anhydrous AlCl3] B ->[acid KMnO4] C}\]
Complete the following reaction.
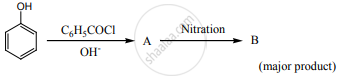
Phenol is distilled with Zn dust followed by Friedel-crafts alkylation with propyl chloride to give a compound B, B on oxidation gives (c) Identify A, B and C.
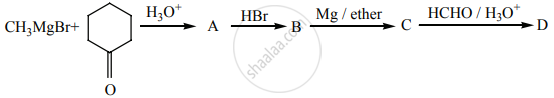
Identify A, B, C, D and write the complete equation.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
What will be the product (X and A) for the following reaction.
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
