उत्तर सहित प्रश्न बैंक
- बालभारती उत्तर (महाराष्ट्र)
- समचेर कालवी उत्तर (तमिलनाडु)
- एनसीईआरटी उत्तर
- आरडी शर्मा उत्तर
- आरडी शर्मा कक्षा १० उत्तर
- आरडी शर्मा कक्षा ९ उत्तर
- लखमीर सिंग उत्तर
- टीएस ग्रेवाल उत्तर
- आईसीएसई कक्षा १० उत्तर
- सेलिना आईसीएसई कॉनसाइस उत्तर
- फ्रँक आईसीएसई उत्तर
- एमएल अग्रवाल उत्तर
- एनसीईआरटी उत्तर कक्षा १२ गणित
- एनसीईआरटी उत्तर कक्षा १२ भौतिक विज्ञान
- एनसीईआरटी उत्तर कक्षा १२ रसायन विज्ञान
- एनसीईआरटी उत्तर कक्षा १२ जीवविज्ञान
- एनसीईआरटी उत्तर कक्षा ११ गणित
- एनसीईआरटी उत्तर कक्षा ११ भौतिक विज्ञान
- एनसीईआरटी उत्तर कक्षा ११ रसायन विज्ञान
- एनसीईआरटी उत्तर कक्षा ११ जीवविज्ञान
- एनसीईआरटी उत्तर कक्षा १० गणित
- एनसीईआरटी उत्तर कक्षा १० विज्ञान
- एनसीईआरटी उत्तर कक्षा ९ गणित
- एनसीईआरटी उत्तर कक्षा ९ विज्ञान
- सीबीएसई अध्ययन सामग्री
- महाराष्ट्र स्टेट बोर्ड अध्ययन सामग्री
- तमिलनाडु राज्य बोर्ड अध्ययन सामग्री
- सीआईएससीई / इसीसीएसई अध्ययन सामग्री
- मुंबई विश्वविद्यालय इंजीनियरिंग अध्ययन सामग्री
प्रश्न पत्र एवं उत्तर
- सीबीएसई पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ कला
- सीबीएसई पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ वाणिज्य
- सीबीएसई पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ विज्ञान
- सीबीएसई पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १०
- महाराष्ट्र स्टेट बोर्ड पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ कला
- महाराष्ट्र स्टेट बोर्ड पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ वाणिज्य
- महाराष्ट्र स्टेट बोर्ड पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ विज्ञान
- महाराष्ट्र स्टेट बोर्ड पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १०
- सीआईएससीई / इसीसीएसई बोर्ड पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ कला
- सीआईएससीई / इसीसीएसई बोर्ड पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ वाणिज्य
- सीआईएससीई / इसीसीएसई बोर्ड पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १२ विज्ञान
- सीआईएससीई / इसीसीएसई बोर्ड पिछले वर्ष के प्रश्न पत्रों के उत्तर सहित कक्षा १०
© 2026 Shaalaa.com | Contact Us | Privacy Policy
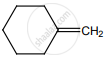
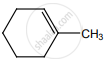
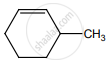
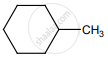
 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives