Advertisements
Advertisements
प्रश्न
Name two colourless metal ions.
Advertisements
उत्तर १
Na+, Ca2+
उत्तर २
Potassium ion (K+), Sodium ion (Na+)
APPEARS IN
संबंधित प्रश्न
When a metallic oxide is dissolved in water, the solution formed has a high concentration of ____________ ions (H+, h3O+, OH-)
Choose the most appropriate answer from the following list of oxides which fit the description.
[SO2, SiO2, Al2O3, MgO, CO, Na2O]
A basic oxide.
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
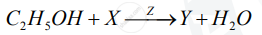
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Give a chemical test to distinguish between the given pairs of chemicals:
Lead nitrate solution and Zinc nitrate solution
Name a weak alkali.
Give two examples of amphoteric hydroxides.
Name the chloride of a metal which is soluble in excess of ammonium hydroxide.
Fill in the blank.
The hydroxide which is soluble in excess of NaOH is _________ [Zn(OH)2 / Fe(OH)3 / Fe(OH)2].
Fill in the blank.
The substance/s which react with hot conc. NaOH solution and undergoes a neutralization reaction ______ [Al2O3 / Al / Al(OH)3]
Write balanced equations for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
