Advertisements
Advertisements
प्रश्न
Name the metal hydroxides which are :
(a) Insoluble (b) Soluble in
(i) Caustic soda solution (ii) Ammonium hydroxide solution.
Advertisements
उत्तर
|
Name of solution |
Soluble metal hydroxides |
Insoluble metal hydroxides |
|
Caustic soda solution |
Zn(OH)2 Pb(OH)2 |
Fe(OH)3 |
|
Ammonium hydroxide solution |
Zn(OH)2 Cu(OH)2 |
Fe(OH)3 Fe(OH)2 |
संबंधित प्रश्न
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
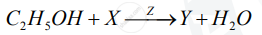
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Name a metallic oxide soluble in excess of caustic soda solution.
Write balanced equation for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
Name a coloured metallic oxide which dissolves in alkalis to yield colourless solutions.
Name a compound containing zinc in the anion.
Choose the correct answer from the options given below :
Metal + alkali → salt + _________
Write balanced equations for a metal that evolves into a gas which burns with a pop sound when boiled with alkali solutions.
Write balanced equations for a metal that evolves into a gas which burns with a pop sound when boiled with alkali solutions.
Write balanced equations for a Metal that Evolves a Gas Which Burns with a Pop Sound When Boiled with Alkali Solutions.
Write balanced equations for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
