Advertisements
Advertisements
प्रश्न
Match List I with List II:
| List I | List II |
| Coke | Carbon atoms are sp3 hybridized. |
| Diamond | Used as a dry lubricant. |
| Fullerene | Used as a reducing agent. |
| Graphite | Cage like molecules |
Choose the correct answer from the options given below:
विकल्प
A - IV, B - I, C - II, D - III
A - III, B - I, C - IV, D - II
A - III, B - IV, C - I, D - II
A - II, B - IV, C - I, D - III
MCQ
जोड़ियाँ मिलाइएँ
Advertisements
उत्तर
A - III, B - I, C - IV, D - II
Explanation:
| List I | List II |
| Coke | Used as a reducing agent. |
| Diamond | Carbon atoms are sp3 hybridized. |
| Fullerene | Cage like molecules |
| Graphite | Used as a dry lubricant. |
Coke: It is used as a reducing agent in carbon reduction methods (in metallurgical process).
Diamond: It is an allotrope of carbon in which each carbon is sp3 hybridized.
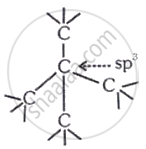
Fullerene: It contains pentagonal & hexagonal rings (cage like structure).
Graphite: It is a soft solid because graphite layers are bonded with weak Vander Waals attractions.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
