Advertisements
Advertisements
प्रश्न
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
Figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00 eV, E2 = 1.78 eV and E3 = 2.95 eV. The ground state is considered 0 eV for reference.
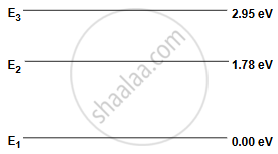
What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
संख्यात्मक
Advertisements
उत्तर
Given, E1 = 0 eV, E2 = 1.78 eV
h = 6.6 × 10−34 J s, c = 3 × 108 m s−1
1 eV = 1.6 × 10−19 J
ΔE = E2 − E1
= 1.78 − 0
= 1.78 eV
Using, E = `(hc)/lambda`
`lambda = (hc)/(\DeltaE)`
= `(6.6 xx 10^-34 xx 3 xx 10^8)/(1.78 xx 1.6 xx 10^-19)`
= 6.95 × 10−7 m
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
