Advertisements
Advertisements
प्रश्न
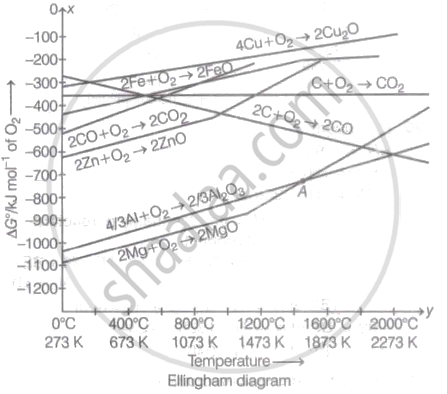
How many moles of gaseous oxygen at one atmosphere is considered for the reaction with element for plotting a graph in Ellingham diagram?
विकल्प
0.5
2
1
0.25
MCQ
Advertisements
उत्तर
1
Explanation:
The standard free energy change (6G0 ) per 1 mole of oxygen is represented on the y-axis, while temperature (K) is plotted on the x-axis in the Ellingham diagram graph. For the Ellingham diagram, one mole of gaseous oxygen at one atmosphere is used to create a graph for the reaction with element. The following is an example:
shaalaa.com
Introduction of States of Matter: Gaseous and Liquid States
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
