Advertisements
Advertisements
प्रश्न
How can we enhance the reactivity of aryl halides?
Advertisements
उत्तर
The reactivity of aryl halides can be increased by the presence of an electron withdrawing group (-NO2) at ortho and para positions. No effect is observed by the presence of electron withdrawing group at weta-position Mechanism of the reaction is as depicted with OH ion:
The presence of NO2 groups at ortho and para positions withdraws electrons density from the benzene ring and therefore, facilitates the attack of the nucleophile on haloarenes. The carbanion, thus formed is stabilized through resonance as shown below:

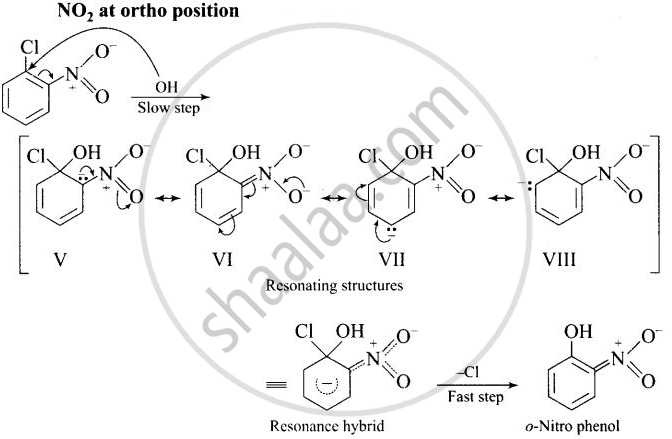
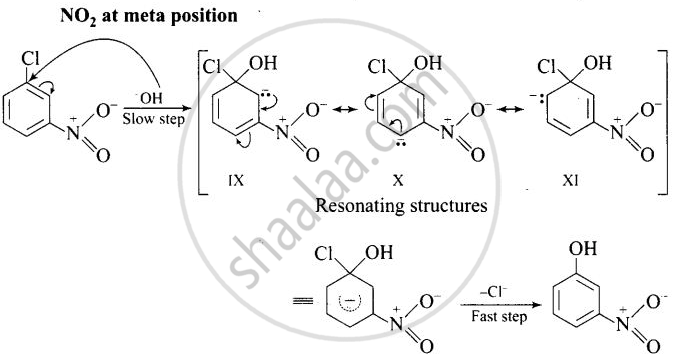
It is clear from above structure that in case of o- and p-chlorobenzenes, on the resonating structures (II in case of p-nitro chlorobenzene and V in case of o-chlorobenzene) bear a negative charge on the carbon atom bearing the \[\ce{NO2}\] group.
Therefore, these carbanions are stabilized by the -NO2 groups as well as π-electrons of the benzene ring. However, in case of m-nitrochlorobenzene, none of the resonating structures bear the negative charge on carbon atom bearing the -NO2 group. Therefore, the nitro group at meta position does not stabilize the negative charge but the carbanion is stabilized only by the p-electrons of the benzene ring. In other words, the carbanions formed from o-nitrochlorobenzene and p-nitrochlorobenzene are most stable than that formed from m-nitrochlorobenzcne.
Thus, the presence of electron withdrawing groups at o- and p-positions (but not at m-positions) w.r.t. halogen atom activates the aryl halides towards nucleophilic substitution reaction.
