Advertisements
Advertisements
प्रश्न
Given below are two statements:
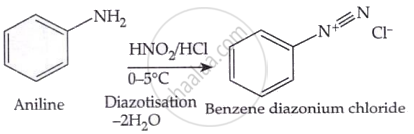
Statement I: Benzenediazonium salt is prepared by the reaction of aniline with nitrous acid at 273-278 K. It decomposes easily in the dry state.
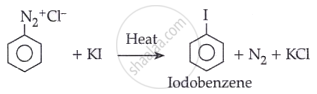
Statement II: Insertion of iodine into the benzene ring is difficult and hence, iodobenzene is prepared through the reaction of benzenediazonium salt with KI.
In the light of the above statements, choose the most appropriate answer from the options given below:
विकल्प
Both Statement I and Statement II are correct.
Both Statement I and Statement II are incorrect.
Statement I is correct but Statement II is incorrect.
Statement I is incorrect but Statement II is correct.
Advertisements
उत्तर
Both Statement I and Statement II are correct.
Explanation:
Aniline and nitrous acid are reacted at a temperature range of 273-278 Kelvin (0-5 degrees Celsius) to produce benzene diazonium salt, more precisely, benzenediazonium chloride. Primary aromatic amines are transformed into diazonium salts by a process known as diazotisation. The diazonium salt is unstable and is not usually preserved since it breaks down quickly in the dry condition.
Iodine is difficult to introduce into the benzene ring; hence, iodobenzene is normally synthesised by reacting benzenediazonium salt with potassium iodide (KI).