Advertisements
Advertisements
प्रश्न
Give two reasons for use of mercury as a barometric liquid.
Advertisements
उत्तर
Two advantages of using mercury as barometric liquid:
(i) The density of mercury is greater than that of all the liquids, so only 0.76m height of mercury column is needed to balance the normal atmospheric pressure.
(ii) The mercury neither wets nor sticks to the glass tube therefore it gives the correct reading.
APPEARS IN
संबंधित प्रश्न
State two experiences that made you think that air exerts pressure.
Give reason for the following :
Two holes are made in a sealed oil tin to take out oil from it.
Give reasons for the following:
Temperature and pressure are inversely related to one another.
State briefly, how and why the atmospheric pressure of a place varies with the altitude. Draw an approximate graph to illustrate this variation.
Why do not we feel uneasy even under enormous pressure of the atmosphere above as?
How is the reading of a barometer affected when it is taken to: A hill
The standard air pressure at sea level is ______.
The small variations in pressure that exist largely determine the ______ and ______ patterns of the earth.
It is much easier to burst an inflated balloon with a needle than by a finger. Explain.
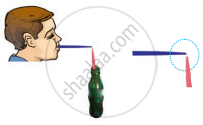
Figure shows a child blowing air with a straw near the opening of another straw which has its other end in a soft drink bottle. It was observed that the level of the soft drink in the straw rises up as soon as air is blown over its open end. Which one of the following best explains the reason for rise in level of the drink?