Advertisements
Advertisements
प्रश्न
Give the structure and name of the product in the following reaction.
Chlorination of toluene in the presence of anhydrous AlCl3.
रासायनिक समीकरण/संरचनाएँ
Advertisements
उत्तर
This is an example of electrophilic aromatic substitution (EAS). Toluene (C6H5CH3) is an activated benzene ring due to the +I and hyperconjugation effects of the methyl (–CH3) group, which directs incoming electrophiles to the ortho and para positions.
- Reaction Equation:
\[\ce{C6H5CH3 + Cl2 ->[AlCl3][] \underset{o-chlorotoluene}{o-ClC6H4CH3} + \underset{p-chlorotoluene}{p-ClC6H4CH3} + HCl}\] - Structures of the Product:
- Ortho-chlorotoluene
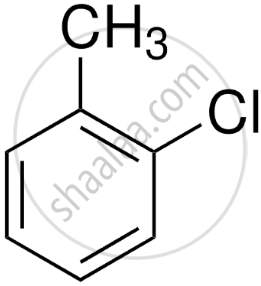
- Para-chlorotoluene

- Ortho-chlorotoluene
- Name of Product:
- Ortho-chlorotoluene (1-chloro-2-methylbenzene)
Chlorine at the position next to the methyl group. - Para-chlorotoluene (1-chloro-4-methylbenzene) (major product due to less steric hindrance)
- Ortho-chlorotoluene (1-chloro-2-methylbenzene)
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
