Advertisements
Advertisements
प्रश्न
Fill in the cross word given in figure with the help of the clues provided.
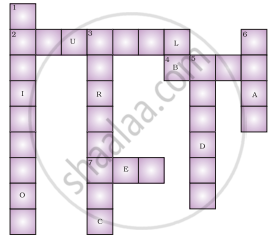
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour in basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Advertisements
उत्तर
Across
2. Neutral
4. Base
7. Red
Down
1. Indicator
3. Turmeric
5. Acidic
6. Heat
APPEARS IN
संबंधित प्रश्न
Name the source from which litmus solution is obtained. What is the use of this solution?
How will you test for the gas which is liberated when hydrochloric acid reacts with an active metal?
Give the names and formulae of two strong acids.
What happens during a wasp sting? What is its remedy?
Write any two important uses of bases.
Name the acid present in vinegar.
Name an acid used to remove ink spots.
Which one of the following acids undergoes complete ionisation?
Bring the soil sample of your area, find out if it is acidic, basic or neutral. Discuss with farmers if they treat the soil in any manner.
In a set of the given reactions, acetic acid yielded a product C.
\[\begin{array}{cc}
\phantom{}\ce{\underset{}{CH3COOH + PCl5} ->[][ ] \underset{}{A}->[C6H6][anhy.AlCl3 ] \underset{}{B}->[C2H5MgBr][Ether] \underset{}{C}}\\
\end{array}\]
Product C would be:
