Advertisements
Advertisements
प्रश्न
Explain why dilute hydrochloric acid cannot be concentrated by boiling beyond 22.2%.
Advertisements
उत्तर
Distillation slowly increases the strength of a weak hydrochloric acid solution until it has 22.2% HCl by weight and boils at 110°C. Once this level is reached, boiling cannot be used to make the acid stronger anymore. When this concentration is attained, boiling [constant boiling mixture or azeotrope] cannot be used to enhance the acid concentration any further.
Water and HCl are the main components of vapours that form before 110°C, although HCl molecules make up the majority of vapours that form above this temperature.
APPEARS IN
संबंधित प्रश्न
State your observations when Dilute Hydrochloric acid is added to Copper carbonate
State your observation When moist starch iodide paper is introduced into chlorine gas.
Give a chemical test to distinguish between the given pairs of chemicals:
Sodium chloride solution and Sodium nitrate solution
What is the function of HCI in preparation of aqua-regia?
What must be added to sodium chloride to prepare hydrogen chloride?
How will you identify?
An amphoteric hydroxide
Select from the list given (a to e) one substances in each case which matches the description given in parts (i) to (v). (Note : Each substance is used only one in the answer)
(a) Nitroso Iron (II) Sulphate
(b) Iron (III) chloride
(c) Chromium sulphate
(d) Lead (II) chloride
(e) Sodium chloride
(i) A compound which is deliquescent
(ii) A compound which is insoluble in cold water, but soluble in hot water
(iii) The compound responsible for the brown ring during the brown ring test of nitrate iron
(iv) A compound whose aqueous solution is neutral in nature
(v) The compound which is responsible for the green colouration when sulphur dioxide is passed through acidified potassium dichromate solution
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
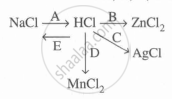
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
