Outline the principle of refining of metal by the following method:
Zone refining
Explain the zone refining process with an example.
Explain zone refining.
Outline the principle of refining of metal by the following method:
Zone refining
When a metal with a high degree of purity is needed, this technique is employed. For instance, this process refines silicon, gallium, and germanium, which are utilised as semiconductors. The technique is predicated on the observation that an impurity’s solubility varies depending on whether the metal to be purified is liquid or solid.
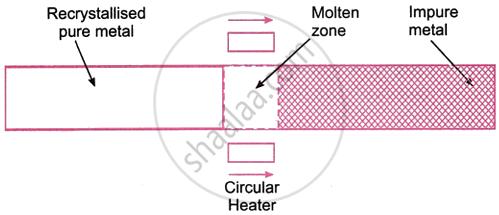
The impure metal is cast into a thin rod using this method. This pole has a round heater attached to it. Slowly, the heater is moved along the rod. The rod melts at the heated zone, and while the heater continues, the impurities move to the nearby molten zone, causing the remaining pure metal to recrystallize. As the heater travels along the rod, the impurities continue to flow from one zone to the next until they are eventually gathered at the other end of the rod. After a number of repetitions, the end that contains the contaminants is finally chopped off. The metal that makes up the remaining rod is extremely pure.
Extraction of gold and silver involves leaching with cyanide ion. silver is later recovered by
The following set of reactions are used in refining Zirconium
\[\ce{Zr (impure) + 2I2 ->[523 K] ZrI4}\]
\[\ce{ZrI4 ->[1800 K] Zr (pure) + 2I2}\]
This method is known as
In the electrolytic refining of copper, which one of the following is used as anode?
Describe a method for refining nickel.
Give the basic requirement for vapour phase refining.
Describe the role of the following in the process mentioned.
Cryolite in the extraction of aluminium.
Describe the role of the following in the process mentioned.
Iodine in the refining of Zirconium.
Explain the principle of electrolytic refining with an example.