Advertisements
Advertisements
प्रश्न
Explain the smelting process.
Advertisements
उत्तर
Smelting (in a Blast Furnace): The charge consisting of roasted ore, coke, and limestone in the ratio 8 : 4: 1 is smelted in a blast furnace by introducing it through the cup and cone arrangement at the top. There are three important regions in the furnace.
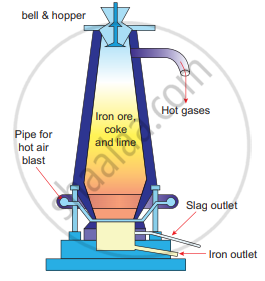
Blast Furnace
(a) The Lower Region (Combustion Zone): The temperature is at 1500°C. In this region, coke bums with oxygen to form CO2 when the charge comes in contact with a hot blast of air.
\[\ce{C + O2->[1500°C][\Delta]CO2 + Heat}\]
It is an exothermic reaction since heat is liberated.
(b) The Middle Region (Fusion Zone): The temperature prevails at 1000°C. In this region, CO2 is reduced to CO.
\[\ce{CO2 + C ->[1000°C][\Delta]2CO - Heat}\]
Limestone decomposes to calcium oxide and CO2
\[\ce{CaCO3->[][\Delta]CaO + CO2 - Heat}\]
These two reactions are endothermic due to the absorption of heat. Calcium oxide combines with silica to form calcium silicate slag.
CaO + SiO2 → CaSiO3
(c) The Upper Region (Reduction Zone): The temperature prevails at 400°C. In this region, carbon monoxide reduces ferric oxide to form a fairly pure spongy iron.
\[\ce{Fe2O3 + 3CO->[400°C]2Fe + 3CO2}\]
The molten iron is collected at the bottom of the furnace after removing the slag.
The iron thus formed is called pig iron. It is remelted and cast into different molds. This iron is called cast iron.
APPEARS IN
संबंधित प्रश्न
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: carbonates
A student has been collecting silver coins and copper coins. One day she observed a black
coating on silver coins and a green coating on copper coins. Which chemical phenomenon is
responsible for these coatings? Write the names of black and green coatings.
Name:
(a) the processes involved in
concentration :-
Explain the following terms:
(c) slag
Give the ionization reactions of electrolyte used in Hall’s process. write the reaction at the cathode and the anode. Why the anode has to be replaced in this process?
Where are the cathode and anode in the electrolytic cell? Name the material used for these?
Explain with reason:
Why is powdered coke sprinkled on top of the electrolyte?
A compound that is added to lower the fusion temperature of the electrolytic bath in the extraction of aluminium.
Draw a neat and labelled diagram for Froth Floatation Process.
A process of extracting metals from aqueous solutions of their salts using suitable reducing agents is called ______
