Advertisements
Advertisements
प्रश्न
Explain the following processe with a suitable example in the given case:
Chain-growth polymerisation
Give an example of chain growth polymerisation.
Advertisements
उत्तर
Through a chain reaction process involving specific reactive intermediates, chain growth polymerization occurs. The mechanism entails a sequential process.
Initiation: An initiator that can create a reactive intermediate, like a free radical (or a carbocation, or a carbanion), is necessary for chain-growth polymerization. For this, organic peroxides like benzoyl peroxide are typically used. A small quantity of initiator is provided, and it is broken down by heat, light, or an oxidation-reduction reaction to create reactive species, such as free radicals. In order to create a reactive intermediate monomer with an active center, the free radical attacked a monomer molecule.
\[\ce{\underset{Organic peroxide}{R - CO - O - O - OC - R} ->[Heat] 2R - CO - \overset{\bullet}{O} -> \underset{Free radical}{2R^\bullet} + 2CO2 R^\bullet + \underset{Ethylene}{H2C = CH2} -> \underset{(reactive intermediate)}{\underset{active centre}{\underset{Monomer with}{R - CH2 - \overset{\bullet}{C}H2}}}}\]
(R = CH3 or C6H5 group)
Chain propagation: Now that the monomer has an active center, it attacks other monomer molecules to create dimer, trimer, and so on polymer units with active centers (free radicals).
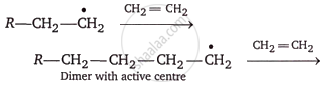
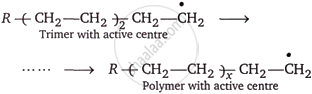
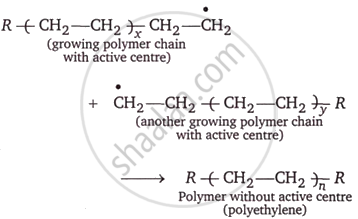
Chain termination: A specific mechanism that involves removing the active center from the polymer chain stops its growth.
Notes
Students should refer to the answer according to their questions.
