Advertisements
Advertisements
प्रश्न
Draw resonance hybrid structure of SO2 in two canonical forms.
Advertisements
उत्तर
Resonance hybrid structure of SO2:
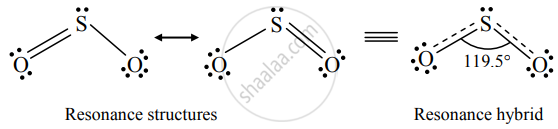
APPEARS IN
संबंधित प्रश्न
Ozone layer is depleted by ______.
Answer the following.
Give one example showing reducing property of ozone.
What happens when lead sulfide reacts with ozone \[\ce{O3}\]?
Discuss the structure of sulfur dioxide.
Draw structures of XeF6.
Answer the following.
Draw structures of XeOF4
Answer the following.
Draw structures of XeF2.
What is the O−S−O bond angle in SO2?
Complete the following reaction:
\[\ce{SO_{2(g)} + Cl_{2(g)} ->[Charcoal]}\] ?
Name the solution which is formed by passing sulfur dioxide in water.
Select the INCORRECT statement.
Which of the following molecule does not contain oxygen?
Which of the following is also called as nitrogen sesquioxide?
Find the CORRECT statement.
\[\ce{I2_{(g)} + H2S_{(g)} -> 2HI_{(g)} + S_{(s)}}\]
Which among the following oxides is amphoteric in nature?
Which one is amphoteric oxide?
An element having electronic configuration 1s2 2s2 2p6 3s2 3p6 4s1 forms ______.
High concentration of which of the following in the atmosphere leads to stiffness of flower buds which eventually fall off from plants?
What will be the formula of an oxide of iodine (atomic mass = 127) which contains 25.4 g of iodine and 8g of oxygen?
Give reason:
Bleaching action of ozone is also called dry bleach.
What are the basic oxides? Explain with an example.
Mention the value of the bond angle in SO2.
Electrolytic method of preparation of dioxygen.
What is oxide?
