Advertisements
Advertisements
प्रश्न
Discuss the structures of the hydrides of group 15 elements.
Advertisements
उत्तर
The sp3-hybridisation of the central atom, M, creates the covalent hydrides (MH3, \[\ce{M = N}\], P, As, Sb, or Bi) of group 15 elements. For example, the electronic configuration of the central N atom in NH3 is \[\ce{1s^2 2s^2 2p^1_x 2p^1_y 2p^1_z}\]. In order to create four hybrid orbitals of equal energy that are orientated toward the four corners of a regular tetrahedron at an angle of 109°28', the fully filled 2s and the three half-filled 2p-orbitals of nitrogen atoms undergo sp3 hybridisation during the synthesis of NH3.
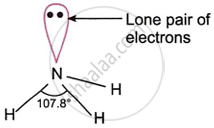
While the fourth hybrid orbital contains a single pair of electrons and is non-bonding, the three singly occupied hybrid orbitals overlap with the 1s-atomic orbitals of three hydrogen atoms to produce three \[\ce{N - H}\] σ-bonds. As shown in the figure, an NH3 pyramidal molecule is thus created. The three \[\ce{N - H}\] bond pairs are attracted by the single pair of electrons on the central nitrogen atom.
The \[\ce{H - N - H}\] bond angle reduces from the predicted value of 109°28' to 107.8° because the lone pair-bond pair repulsion is larger than the bond pair-bond pair repulsion. As a result, the ammonia molecule takes on the shape of a distorted pyramid with an \[\ce{H - N - H}\] bond angle of 107.8°.
