Advertisements
Advertisements
प्रश्न
Differentiate between metals and non-metals with reference to –
(a) number of electrons in outer or valence shell
(b) formation of cation and anion
(c) reaction with dilute acids.
अंतर स्पष्ट करें
Advertisements
उत्तर
| Chemical Property | Metals | Non-metals |
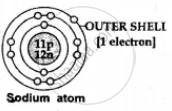
| (a) No.of electrons in outer shell |
1, 2 or 3 electrons -
|
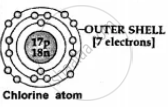
4, 5, 6 or 7 electrons - in the outer or valence shell of a non-metal
|
| (b) Ion formation | Metal lose or donate valence electrons and form cations [positive ions] e.g. Na - 1e- → Na1+ [caution] |
Non-metal accept valence electrons and form anions [negative ions] e.g. Cl + 1e- → Cl1- [anion] |
| (c) Reaction with acids | React with dilute acids to give hydrogen | Do not react with dilute acids to give hydrogen. |
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?