Advertisements
Advertisements
प्रश्न
Describe vertical retort process used for the extraction of zinc.
Advertisements
उत्तर
In the vertical retort process, roasted ore is combined with powdered coal or coke and heated in a retort.
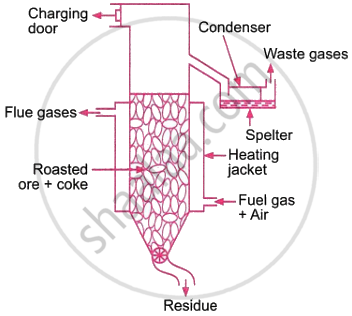
The retort is covered by a jacket that burns a mixture of fuel gas and air. The heat produced heats the retort to around 1600 K. Zinc oxide is converted to zinc.
\[\ce{ZnO + C ->[1600 K] Zn + Co}\]
As a result, zinc is vaporised. The CO and zinc vapour are extracted and condensed from an exit close to the retort’s top. Zinc spelter is the term for the condensed zinc that is so produced. Carbon monoxide escapes instead of condensing. A screw conveyer installed in the bottom removes the residue once the new charge is introduced through the charging door. As a result, the process never stops.
