Advertisements
Advertisements
प्रश्न
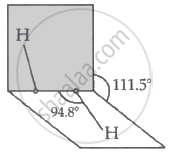
Assertion A: The dihedral angles in H2O2 in gaseous phase is 90.2° and in solid phase is 111.5°.
Reason R: The change in dihedral angle in solid and gaseous phase is due to the difference in the intermolecular forces.
Choose the most appropriate answer from the options given below for A and R.7
विकल्प
Both A and R are correct but R is not the correct explanation of A.
A is correct but R is not correct.
Both A and R are correct and R is the correct explanation of A.
A is not correct but R is correct.
Advertisements
उत्तर
A is not correct but R is correct.
Explanation:
H2O2 and water combine in all amounts and have a structure that is somewhat comparable. Since hydrogen bonding in H2O2 affects the dihedral angle, H2O2 in the gas phase has a somewhat different structure from H2O2 in the solid phase.
The given images show structure of H2O2 in both gas and solid phase.
 |
 |
| (a) Gas phase | (b) Solid phase |
The structure of hydrogen peroxide is non- planar. The dihedral angle in gas phase 111.5° whereas, in solid phase the dihedral angle is 90.2°. H2O2 occurs in solid phase at 110 K.
The structure of H2O2 in gas phase is such that to minimize the repulsion between lone pairs and the O-H bond pairs. H2O2 structure is similar to that of an open book consisting 2 planes, such that each plane has one O-H bond pair.
H2O2 exhibits a skewed structure in both its solid and gaseous states due to the repulsion between its - OH bonds and the lone pairs of electrons on each of its O atoms. As a result, the molecule exhibits impeded rotation around a single bond. As hydrogen bonds weaken in the solid phase, the dihedral angle also decreases.
Hence, assertion A is incorrect but, reason R is correct.
