Advertisements
Advertisements
प्रश्न
Answer the following question:
What do you understand by the statement: ‘metals are ductile and malleable’?
Advertisements
उत्तर
Metals are ductile, i.e., they can be drawn or stretched into thin wires.
They are malleable, i.e., they can be beaten into thin sheets.
APPEARS IN
संबंधित प्रश्न
Tick the most appropriate answer
Solutions are
- heterogeneous mixtures.
- compounds
- homogeneous mixtures.
- elements
Match the columns.
| column A | column B |
|
1. to separate grain from chaff |
a. filtration |
| 2. to saparate sawdust from water | b. sedimentation and deeantation |
| 3. to separate iodine from sodium chloride | c. winnowing |
| 4. to separate iron fillings from sand | d.sublimation |
|
5. it is used to separate sand and water |
e maganetic separation |
|
|
f .handpicking |
Why is it necessary to separate the constituents of a mixture.
Give two examples of the following.
Inert gases
Match the Columns:
| Column A | Column B | ||
| (a) | Metals | (i) | Non-Reactive |
| (b) | Molecules | (ii) | Brittle |
| (c) | Non-Metals | (iii) | Lustrous |
| (d) | Noble Gases | (iv) | Smallest unit of Compound |
Write the names of elements from the following symbol:
Fe
Select the correct answer to the statement given below:
A non-metal which exists in the liquid state.
Explain the term ‘mixture’.
Higher Order Thinking Skills.
In the diagram given below, the circle, square and triangle represent the atoms of different elements.
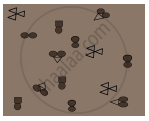
Identify all combinations that represent
- molecule of a compound
- molecule of an element consisting of two atoms
- molecule of an element consisting of three atoms
How are elements classified?
