Advertisements
Advertisements
प्रश्न
Answer the following
How will you illustrate the principle, minimization of steps?
Advertisements
उत्तर
Reduce derivatives (Minimization of steps):
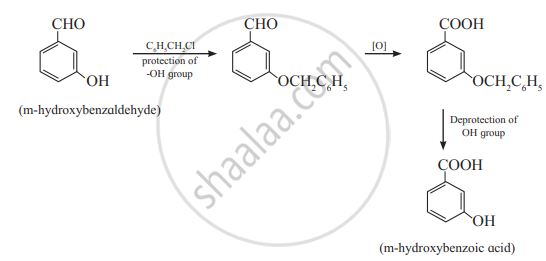
In organic synthesis protecting or blocking groups is commonly used. According to this principle of green chemistry, unnecessary derivatization, for example, installation/removal of the use of protecting groups should be minimized or avoided if possible, because such steps require additional reagents and can generate waste.
Illustration:
a. In organic synthesis, the protection of some functional groups is required. Again, the deprotection of the functional group is required at the end.
For example: Synthesis of m-hydroxybenzoic acid from mhydroxybenzaldehyde.
b. In such cases, the atom economy is also less.
c. The green chemistry principle aims to develop the methodology where unnecessary steps should be avoided. This can be done if possible, by using practicable biocatalytic reactions, which very often need no protection of the selective group.
APPEARS IN
संबंधित प्रश्न
Name the γ-isomer of BHC.
Explain any three principles of green chemistry.
Answer the following:
Explain the atom economy with a suitable example.
Answer the following
Give two examples of catalysts.
Name the γ-isomer of Benzene hexachloride.
Which principle of green chemistry has its perspective towards carrying out reactions at room temperature and pressure?
Define atom economy.
Write one example of safer solvent and hazardous solvent.
Explain prevention of waste or by products which is one of the principles of green chemistry.
____________ can be categorised as a green solvent.
In the manufacture of phenol from isopropyl benzene, the important by-product obtained is ____________.
Drath and Frost developed a green technology for the synthesis of ____________.
The formula for percentage atom economy is:
`("Formula weight of the desired product")/("'X'") xx 100`
Here, 'X' is ____________.
Mandelonitrile is obtained by the reaction between hydrogen cyanide and ______.
The international plastic recycle mark with 'Number 1' is given to ____________.
Which of the following is recycled to prepare foam packaging and light switchplates?
γ-isomer of BHC is called as ____________.
All the following statements are CORRECT, EXCEPT:
Identify the polymer used in making floor tiles.
Define the role of safer chemistry in accident prevention.
Calculate the atom economy of the following:
(At mass of C = 12, H = 1, 0 = 16)
\[\ce{CH3 - CH2 - OH ->[\Delta] CH2 = CH2 + H2O}\]
How many principles does green chemistry have?
Which of the following is used as green solvent?
Which of the following is primary benzylic alcohol?
