Advertisements
Advertisements
प्रश्न
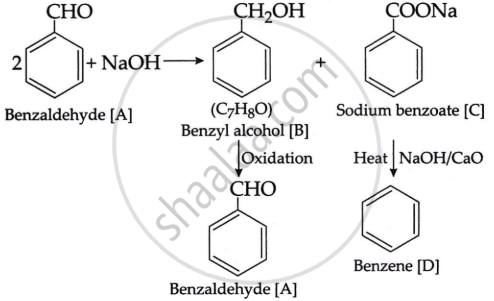
An organic compound [A], having a specific smell forms two compounds [B] and [C] by reacting with conc. sodium hydroxide. The molecular formula of compound [B] is C7H8O, which forms compound [A] again on oxidation. Compound [C] forms benzene on heating with soda lime.
Write the structures of compounds [A], [B] and [C]. Also, write the reactions involved.
Advertisements
उत्तर
The chemical component [A] must be an aromatic aldehyde, i.e., benzaldehyde with a distinct odour. When benzaldehyde is combined with NaOH. It undergoes Cannizzaro's reaction, yielding benzyl alcohol, [B] of the formula C7H8O and a sodium salt of benzoic acid [C]. Benzyl alcohol degrades into benzaldehyde [A] when oxidised. When heated with soda lime, the chemical [C] produces an aromatic hydrocarbon [D], which is benzene. The enters series of reaction is as given: