Advertisements
Advertisements
प्रश्न
A student was instructed by the teacher to prepare and collect ammonia gas in the laboratory by using aluminium nitride. The student had set up the apparatus as shown in the diagram below. Study the given diagram and answer the following questions:
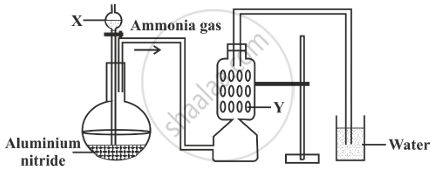
- Name the substance X added through the thistle funnel by the student.
- Write a balanced equation for the reaction occurring between Aluminium nitride and substance X.
- Identify the substance Y.
- State the function of Y.
- Why could the student not collect ammonia gas at the end of the experiment?
रासायनिक समीकरण/संरचनाएँ
एक शब्द/वाक्यांश उत्तर
लघु उत्तरीय
Advertisements
उत्तर
- Warm water
- \[\ce{AlN + 3H2O -> Al(OH)3 + NH3(\uparrow)}\]
- Calcium oxide (CaO)
- Y acts as a drying agent for ammonia gas. Its function is to remove moisture from ammonia gas.
- The student could not collect ammonia gas at the end of the experiment because it is highly soluble in water, dissolving to form aqueous ammonia.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
2024-2025 (March) Official Board
