Advertisements
Advertisements
Rewrite the following reaction by filling the blanks:
\[\ce{CH3 - CH - CH2 + HBr -> \underset{(major)}{\underline{}\underline{}\underline{}\underline{}} + \underset{(minor)}{\underline{}\underline{}\underline{}\underline{}}}\]
Concept: undefined >> undefined
Rewrite the following reaction by filling the blanks:
\[\ce{CH3 - CH = CH2 + HBr ->[peroxide] \underset{(major)}{\underline{}\underline{}\underline{}\underline{}} + \underset{(minor)}{\underline{}\underline{}\underline{}\underline{}}}\]
Concept: undefined >> undefined
Advertisements
Rewrite the following reaction by filling the blanks:
\[\ce{(CH3)2C = CHCH3 + HBr ->[peroxide] \underset{(major)}{\underline{}\underline{}\underline{}\underline{}} + \underset{(minor)}{\underline{}\underline{}\underline{}\underline{}}}\]
Concept: undefined >> undefined
The number of octahedral sites per sphere in ccp structure is ______.
Concept: undefined >> undefined
Formulate the galvanic cell in which the following reaction occurs.
2Al(S) + 3Cu2+(aq) → 2Al3+(aq) + 3Cu(S)
Concept: undefined >> undefined
Derive integrated rate law for zero order reaction.
Concept: undefined >> undefined
What is the effect of denaturation on the structure of proteins?
Concept: undefined >> undefined
Calculate the pH of a buffer solution containing 0.1 M CH3COOH and 0.05 M CH3COONa. Dissociation constant of CH3COOH is 1.8 × 10-5 at 25°C.
Concept: undefined >> undefined
Give two reactions in which acidified K2Cr2O7 is used as an oxidizing agent.
Concept: undefined >> undefined
A voltaic cell consisting of Fe2+(aq)| Fe(s) and Bi3+(aq)| Bi(s) electrodes is constructed. When the circuit is closed, mass of Fe electrode decreases and that of Bi electrode increases.
- Write cell formula .
- Which electrode is cathode and which electrode is anode?
- Write electrode reactions and overall cell reaction.
Concept: undefined >> undefined
Identify the product in the following reaction: 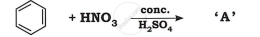
Concept: undefined >> undefined
How are the following compound obtained from alcohol?
Methanal
Concept: undefined >> undefined
How are the following compound obtained from alcohol?
Propanal
Concept: undefined >> undefined
Discuss SN2 mechanism of methyl bromide using aqueous KOH.
Concept: undefined >> undefined
The degree of dissociation of 0.1 M monobasic acid is 0.4%. Its dissociation constant is ______.
Concept: undefined >> undefined
What is ozone umbrella?
Concept: undefined >> undefined
Define the colligative properties.
Concept: undefined >> undefined
Name any two colligative properties.
Concept: undefined >> undefined
