Advertisements
Advertisements
For which one of the following, Bohr model is not valid?
Concept: undefined >> undefined
What is the de Broglie wavelength of an electron of energy 180 eV?
(Mass of electron = 9 x 10-31 kg and Planck's constant = 6.6 x 10-34 Js.)
Concept: undefined >> undefined
Advertisements
The radius of circle, the period of revolution, initial position, and sense of revolution are indicated in the figure.
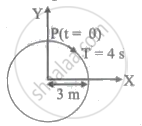
y-projection of the radius vector of rotating particle P is:
Concept: undefined >> undefined
In Bohr's model of hydrogen atom, the period of revolution of the electron in any orbit is proportional to ______.
Concept: undefined >> undefined
In the nth orbit, the energy of an electron `"E"_"n"= -13.6/"n"^2"eV"` for hydrogen atom. The energy required to take the electron from first orbit to second orbit will be ____________.
Concept: undefined >> undefined
When hydrogen atom is in its first excited level, its radius is how many time its ground state radius?
Concept: undefined >> undefined
An iron plate has a circular hole of a diameter 11 cm. Find the diameter of the hole when the plate is uniformly heated from 10° C to 90° C.`[alpha = 12 xx 10^-6//°"C"]`
Concept: undefined >> undefined
According to Bohr's theory, the expression for the kinetic and potential energy of an electron revolving in an orbit is given respectively by ______.
Concept: undefined >> undefined
The binding energy of an electron in nth orbit of the hydrogen atom is given by `"E"_"n" = 13.6/"n"^2 "eV."` The energy required to knock an electron from the second orbit in eV will be ____________.
Concept: undefined >> undefined
A metre scale made of a metal reads accurately at 25 °C. Suppose in an experiment an accuracy of 0.12 mm in 1 m is required, the range of temperature in which the experiment can be performed with this metre scale is ______.(coefficient of linear expansion of the metal is `20 xx 10^-6 / (°"C")`
Concept: undefined >> undefined
A metal sphere 10.01 cm in diameter is placed on a brass ring of internal diameter 10 cm and at the same temperature of 12° C. The temperature up to which they should be heated together so that the metal sphere just passes through the ring is `[alpha_"metal"= 12 xx 10^-6//°"C" and alpha_"brass" =18 xx 10^-6//°"C"]` ____________.
Concept: undefined >> undefined
For an electron, discrete energy levels are characterised by ____________.
Concept: undefined >> undefined
If m is mass of electron, v its velocity, r the radius of stationary circular orbit around a nucleus with charge Ze, then from Bohr's second postulate, the kinetic energy `"K.E." = 1/2"mv"^2` of the electron in C.G.S. system is 2 equal to ____________.
Concept: undefined >> undefined
The unit of intensity of polarization is ____________.
Concept: undefined >> undefined
The average translational kinetic energy of a molecule in a gas becomes equal to 0.49 eV at a temperature about (Boltzmann constant = 1.38 x 10-23 JK-1) ____________.
Concept: undefined >> undefined
Which of the following models was successful in explaining the observed hydrogen spectrum?
Concept: undefined >> undefined
If the speed of an electron of hydrogen atom in the ground state is 2.2 x 106 m/s, then its speed in the third excited state will be ______.
Concept: undefined >> undefined
In hydrogen emission spectrum, for any series, the principal quantum number is n. Corresponding maximum wavelength λ is ______.
(R = Rydberg's constant)
Concept: undefined >> undefined
The acceleration of electron in the first orbit of hydrogen atom is ______.
Concept: undefined >> undefined
For the weight of body of mass 5 kg to be zero on equator of the earth, angular velocity of the earth must be (The radius of earth = 6400 km, acceleration due to gravity = 10 m/s2).
Concept: undefined >> undefined
